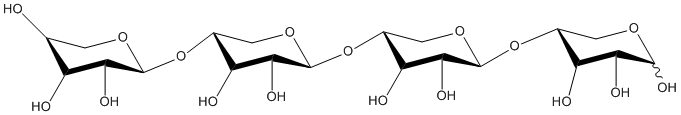
Xylotetraose
General
Type : Glycoside
Chemical_Nomenclature : (2S,3R,4S,5R)-2-[(3R,4R,5R,6S)-6-[(3R,4R,5R,6S)-4,5-dihydroxy-6-[(3R,4R,5R)-4,5,6-trihydroxyoxan-3-yl]oxyoxan-3-yl]oxy-4,5-dihydroxyoxan-3-yl]oxyoxane-3,4,5-triol
Canonical SMILES : C1C(C(C(C(O1)OC2COC(C(C2O)O)OC3COC(C(C3O)O)OC4COC(C(C4O)O)O)O)O)O
InChI : InChI=1S\/C20H34O17\/c21-5-1-32-18(14(27)9(5)22)36-7-3-34-20(16(29)11(7)24)37-8-4-33-19(15(28)12(8)25)35-6-2-31-17(30)13(26)10(6)23\/h5-30H,1-4H2\/t5-,6-,7-,8-,9+,10+,11+,12+,13-,14-,15-,16-,17?,18+,19+,20+\/m1\/s1
InChIKey : KPTPSLHFVHXOBZ-BIKCPUHGSA-N
Other name(s) : beta-D-xylopyranosyl-(1->4)-beta-D-xylopyranosyl-(1->4)-beta-D-xylopyranosyl-(1->4)-D-xylopyranose, D-xylotetraose, CHEBI:62972
MW : 546.5
Formula : C20H34O17
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
Families : A85-Feruloyl-Esterase, Tannase, Lipase_3
References (3)
| Title : Enzymatically-synthesized xylo-oligosaccharides laurate esters as surfactants of interest - Gerard_2020_Carbohydr.Res_495_108090 |
| Author(s) : Gerard D , Meline T , Muzard M , Deleu M , Plantier-Royon R , Remond C |
| Ref : Carbohydr Res , 495 :108090 , 2020 |
| Abstract : Gerard_2020_Carbohydr.Res_495_108090 |
| ESTHER : Gerard_2020_Carbohydr.Res_495_108090 |
| PubMedSearch : Gerard_2020_Carbohydr.Res_495_108090 |
| PubMedID: 32807358 |
| Title : A thermostable feruloyl-esterase from the hemicellulolytic bacterium Thermobacillus xylanilyticus releases phenolic acids from non-pretreated plant cell walls - Rakotoarivonina_2011_Appl.Microbiol.Biotechnol_90_541 |
| Author(s) : Rakotoarivonina H , Hermant B , Chabbert B , Touzel JP , Remond C |
| Ref : Applied Microbiology & Biotechnology , 90 :541 , 2011 |
| Abstract : Rakotoarivonina_2011_Appl.Microbiol.Biotechnol_90_541 |
| ESTHER : Rakotoarivonina_2011_Appl.Microbiol.Biotechnol_90_541 |
| PubMedSearch : Rakotoarivonina_2011_Appl.Microbiol.Biotechnol_90_541 |
| PubMedID: 21279344 |
| Gene_locus related to this paper: thexy-f2vpd7 |
| Title : Degradation of feruloylated oligosaccharides from sugar-beet pulp and wheat bran by ferulic acid esterases from Aspergillus niger - Ralet_1994_Carbohydr.Res_263_257 |
| Author(s) : Ralet MC , Faulds CB , Williamson G , Thibault JF |
| Ref : Carbohydr Res , 263 :257 , 1994 |
| Abstract : Ralet_1994_Carbohydr.Res_263_257 |
| ESTHER : Ralet_1994_Carbohydr.Res_263_257 |
| PubMedSearch : Ralet_1994_Carbohydr.Res_263_257 |
| PubMedID: 7805053 |
| Gene_locus related to this paper: aspng-faeb , aspni-FAEA |