Triolein
General
Type : Acyl-Glycerol || Triacylglycerol || Fatty acid ester || Water-insoluble medium and long chain ester || Oleate
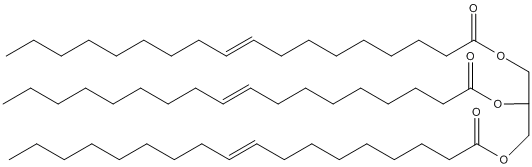
Chemical_Nomenclature : 2,3-bis[[(Z)-octadec-9-enoyl]oxy]propyl (Z)-octadec-9-enoate
Canonical SMILES : CCCCCCCCC=CCCCCCCCC(=O)OCC(COC(=O)CCCCCCCC=CCCCCCCCC)OC(=O)CCCCCCCC=CCCCCCCCC
InChI : InChI=1S\/C57H104O6\/c1-4-7-10-13-16-19-22-25-28-31-34-37-40-43-46-49-55(58)61-52-54(63-57(60)51-48-45-42-39-36-33-30-27-24-21-18-15-12-9-6-3)53-62-56(59)50-47-44-41-38-35-32-29-26-23-20-17-14-11-8-5-2\/h25-30,54H,4-24,31-53H2,1-3H3\/b28-25-,29-26-,30-27-
InChIKey : PHYFQTYBJUILEZ-IUPFWZBJSA-N
Other name(s) : C18:1-triglyceride, Glycerol trioleate, Trioleoylglycerol, Oleic triglyceride, Glycerol triolein, Trioleoylglyceride
MW : 885.43
Formula : C57H104O6
CAS_number :
PubChem :
UniChem :
Iuphar :
References (4)
| Title : Carboxylesterase 2 proteins are efficient diglyceride and monoglyceride lipases possibly implicated in metabolic disease - Chalhoub_2021_J.Lipid.Res__100075 |
| Author(s) : Chalhoub G , Kolleritsch S , Maresch LK , Taschler U , Pajed L , Tilp A , Natmessnig H , Rosina P , Kien B , Radner FPW , Schicho R , Oberer M , Schoiswohl G , Haemmerle G |
| Ref : J Lipid Res , :100075 , 2021 |
| Abstract : Chalhoub_2021_J.Lipid.Res__100075 |
| ESTHER : Chalhoub_2021_J.Lipid.Res__100075 |
| PubMedSearch : Chalhoub_2021_J.Lipid.Res__100075 |
| PubMedID: 33872605 |
| Gene_locus related to this paper: human-CES2 , mouse-Ces2a , mouse-Ces2b , mouse-Ces2c |
| Title : Regulation of hepatic lipase activity by sphingomyelin in plasma lipoproteins - Yang_2015_Biochim.Biophys.Acta_1851_1327 |
| Author(s) : Yang P , Subbaiah PV |
| Ref : Biochimica & Biophysica Acta , 1851 :1327 , 2015 |
| Abstract : Yang_2015_Biochim.Biophys.Acta_1851_1327 |
| ESTHER : Yang_2015_Biochim.Biophys.Acta_1851_1327 |
| PubMedSearch : Yang_2015_Biochim.Biophys.Acta_1851_1327 |
| PubMedID: 26193433 |
| Title : Structure of human pancreatic lipase-related protein 2 with the lid in an open conformation - Eydoux_2008_Biochemistry_47_9553 |
| Author(s) : Eydoux C , Spinelli S , Davis TL , Walker JR , Seitova A , Dhe-Paganon S , de Caro A , Cambillau C , Carriere F |
| Ref : Biochemistry , 47 :9553 , 2008 |
| Abstract : Eydoux_2008_Biochemistry_47_9553 |
| ESTHER : Eydoux_2008_Biochemistry_47_9553 |
| PubMedSearch : Eydoux_2008_Biochemistry_47_9553 |
| PubMedID: 18702514 |
| Gene_locus related to this paper: human-PNLIPRP2 |