Methyl-DEUP
General
Type : Organophosphate || Coumarin
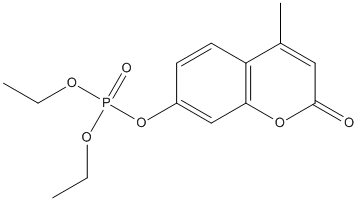
Chemical_Nomenclature : diethyl (4-methyl-2-oxochromen-7-yl) phosphate
Canonical SMILES : CCOP(=O)(OCC)OC1=CC2=C(C=C1)C(=CC(=O)O2)C
InChI : InChI=1S\/C14H17O6P\/c1-4-17-21(16,18-5-2)20-11-6-7-12-10(3)8-14(15)19-13(12)9-11\/h6-9H,4-5H2,1-3H3
InChIKey : GHGRYKAFUNNHBG-UHFFFAOYSA-N
Other name(s) : Diethyl-4-methylumbelliferyl-phosphate, 7-Hydroxy-4-methylcoumarin diethylphosphate, Coumarin, 7-hydroxy-4-methyl-, diethylphosphate, AC1L21T3, ZINC2040214, HE409232, LS-55223
MW : 312.25
Formula : C14H17O6P
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
Families : Carb_B_Arthropoda
References (2)
| Title : Structure and function of an insect alpha-carboxylesterase (alphaEsterase7) associated with insecticide resistance - Jackson_2013_Proc.Natl.Acad.Sci.U.S.A_110_10177 |
| Author(s) : Jackson CJ , Liu JW , Carr PD , Younus F , Coppin C , Meirelles T , Lethier M , Pandey G , Ollis DL , Russell RJ , Weik M , Oakeshott JG |
| Ref : Proc Natl Acad Sci U S A , 110 :10177 , 2013 |
| Abstract : Jackson_2013_Proc.Natl.Acad.Sci.U.S.A_110_10177 |
| ESTHER : Jackson_2013_Proc.Natl.Acad.Sci.U.S.A_110_10177 |
| PubMedSearch : Jackson_2013_Proc.Natl.Acad.Sci.U.S.A_110_10177 |
| PubMedID: 23733941 |
| Gene_locus related to this paper: luccu-E3aest7 |
| Title : Highly sensitive active-site titration of lipase in microscale culture media using fluorescent organophosphorus ester - Fujii_2003_Biochim.Biophys.Acta_1631_197 |
| Author(s) : Fujii R , Utsunomiya Y , Hiratake J , Sogabe A , Sakata K |
| Ref : Biochimica & Biophysica Acta , 1631 :197 , 2003 |
| Abstract : Fujii_2003_Biochim.Biophys.Acta_1631_197 |
| ESTHER : Fujii_2003_Biochim.Biophys.Acta_1631_197 |
| PubMedSearch : Fujii_2003_Biochim.Biophys.Acta_1631_197 |
| PubMedID: 12633686 |