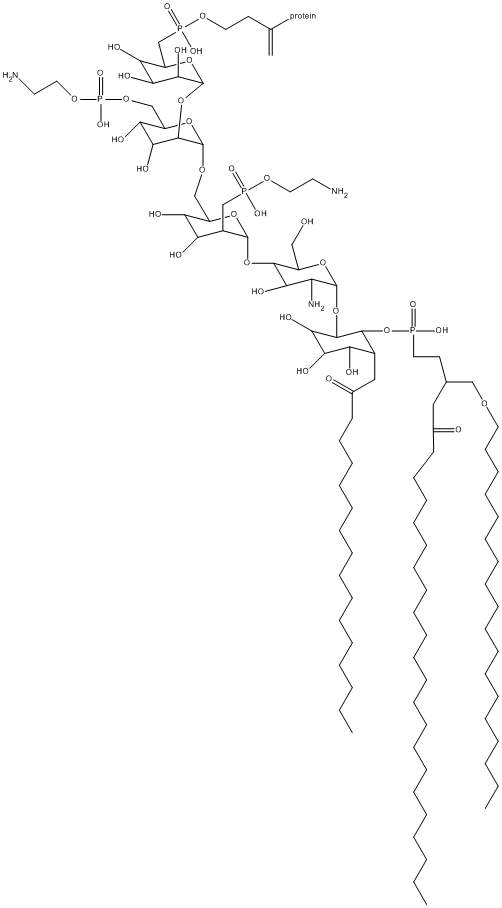
GPI-anchor
General
Type : Phospholipid
Chemical_Nomenclature :
Canonical SMILES :
InChI :
InChIKey :
Other name(s) :
MW :
Formula :
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
Families : PGAP1
References (2)
| Title : Null mutation in PGAP1 impairing Gpi-anchor maturation in patients with intellectual disability and encephalopathy - Murakami_2014_PLoS.Genet_10_e1004320 |
| Author(s) : Murakami Y , Tawamie H , Maeda Y , Buttner C , Buchert R , Radwan F , Schaffer S , Sticht H , Aigner M , Reis A , Kinoshita T , Jamra RA |
| Ref : PLoS Genet , 10 :e1004320 , 2014 |
| Abstract : Murakami_2014_PLoS.Genet_10_e1004320 |
| ESTHER : Murakami_2014_PLoS.Genet_10_e1004320 |
| PubMedSearch : Murakami_2014_PLoS.Genet_10_e1004320 |
| PubMedID: 24784135 |
| Gene_locus related to this paper: human-PGAP1 |
| Title : Inositol deacylation of glycosylphosphatidylinositol-anchored proteins is mediated by mammalian PGAP1 and yeast Bst1p - Tanaka_2004_J.Biol.Chem_279_14256 |
| Author(s) : Tanaka S , Maeda Y , Tashima Y , Kinoshita T |
| Ref : Journal of Biological Chemistry , 279 :14256 , 2004 |
| Abstract : Tanaka_2004_J.Biol.Chem_279_14256 |
| ESTHER : Tanaka_2004_J.Biol.Chem_279_14256 |
| PubMedSearch : Tanaka_2004_J.Biol.Chem_279_14256 |
| PubMedID: 14734546 |
| Gene_locus related to this paper: human-PGAP1 , ratno-q765a7 , yeast-BST1 |