Dabigatran-etexilate
General
Type : Pro-Drug || Not A\/B H target || Drug || Pyridine || Propionate || Benzimidazole
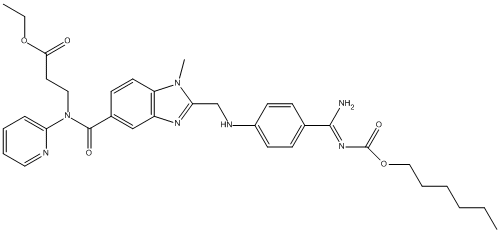
Chemical_Nomenclature : ethyl 3-[[2-[[4-[(Z)-N'-hexoxycarbonylcarbamimidoyl]anilino]methyl]-1-methylbenzimidazole-5-carbonyl]-pyridin-2-ylamino]propanoate
Canonical SMILES : CCCCCCOC(=O)N=C(C1=CC=C(C=C1)NCC2=NC3=C(N2C)C=CC(=C3)C(=O)N(CCC(=O)OCC)C4=CC=CC=N4)N
InChI : InChI=1S\/C34H41N7O5\/c1-4-6-7-10-21-46-34(44)39-32(35)24-12-15-26(16-13-24)37-23-30-38-27-22-25(14-17-28(27)40(30)3)33(43)41(20-18-31(42)45-5-2)29-11-8-9-19-36-29\/h8-9,11-17,19,22,37H,4-7,10,18,20-21,23H2,1-3H3,(H2,35,39,44)
InChIKey : KSGXQBZTULBEEQ-UHFFFAOYSA-N
Other name(s) : Dabigatran etexilate, Pradax, Rendix, Pradaxa, BIBR 1048
Target
Families : Carb_B_Chordata
References (7)
| Title : The Effect of ABCB1 and CES1 Polymorphisms on Plasma Levels of Dabigatran and Risk of Hemorrhagic Complications in Ischemic Stroke Patients - Olserova_2024_Am.J.Ther__ |
| Author(s) : Olserova A , Jansky P , Magerova H , Sramkova T , Kesnerova P , Kmetonyova S , Sulc V , Halmova H , Sramek M , Sarbochova I , Paulasova-Schwabova J , Benesova K , Macek J , Maoska V , Tomek |
| Ref : Am J Ther , : , 2024 |
| Abstract : Olserova_2024_Am.J.Ther__ |
| ESTHER : Olserova_2024_Am.J.Ther__ |
| PubMedSearch : Olserova_2024_Am.J.Ther__ |
| PubMedID: 38525959 |
| Title : Carboxylesterase-2 plays a critical role in dabigatran etexilate active metabolite formation - Laizure_2022_Drug.Metab.Pharmacokinet_47_100479 |
| Author(s) : Laizure SC , Chen F , Farrar JE , Ali D , Yang B , Parker RB |
| Ref : Drug Metab Pharmacokinet , 47 :100479 , 2022 |
| Abstract : Laizure_2022_Drug.Metab.Pharmacokinet_47_100479 |
| ESTHER : Laizure_2022_Drug.Metab.Pharmacokinet_47_100479 |
| PubMedSearch : Laizure_2022_Drug.Metab.Pharmacokinet_47_100479 |
| PubMedID: 36375226 |
| Title : Association between statin use and ischemic stroke or major hemorrhage in patients taking dabigatran for atrial fibrillation - Antoniou_2017_CMAJ_189_E4 |
| Author(s) : Antoniou T , Macdonald EM , Yao Z , Hollands S , Gomes T , Tadrous M , Mamdani MM , Juurlink DN |
| Ref : Cmaj , 189 :E4 , 2017 |
| Abstract : Antoniou_2017_CMAJ_189_E4 |
| ESTHER : Antoniou_2017_CMAJ_189_E4 |
| PubMedSearch : Antoniou_2017_CMAJ_189_E4 |
| PubMedID: 28246253 |
| Title : A Comprehensive Functional Assessment of Carboxylesterase 1 Nonsynonymous Polymorphisms - Wang_2017_Drug.Metab.Dispos_45_1149 |
| Author(s) : Wang X , Rida N , Shi J , Wu AH , Bleske BE , Zhu HJ |
| Ref : Drug Metabolism & Disposition: The Biological Fate of Chemicals , 45 :1149 , 2017 |
| Abstract : Wang_2017_Drug.Metab.Dispos_45_1149 |
| ESTHER : Wang_2017_Drug.Metab.Dispos_45_1149 |
| PubMedSearch : Wang_2017_Drug.Metab.Dispos_45_1149 |
| PubMedID: 28838926 |
| Gene_locus related to this paper: human-CES1 |
| Title : Dabigatran etexilate activation is affected by the CES1 genetic polymorphism G143E (rs71647871) and gender - Shi_2016_Biochem.Pharmacol_119_76 |
| Author(s) : Shi J , Wang X , Nguyen JH , Bleske BE , Liang Y , Liu L , Zhu HJ |
| Ref : Biochemical Pharmacology , 119 :76 , 2016 |
| Abstract : Shi_2016_Biochem.Pharmacol_119_76 |
| ESTHER : Shi_2016_Biochem.Pharmacol_119_76 |
| PubMedSearch : Shi_2016_Biochem.Pharmacol_119_76 |
| PubMedID: 27614009 |
| Gene_locus related to this paper: human-CES1 |
| Title : Impact of endogenous esterase activity on in vitro p-glycoprotein profiling of dabigatran etexilate in caco-2 monolayers - Ishiguro_2014_Drug.Metab.Dispos_42_250 |
| Author(s) : Ishiguro N , Kishimoto W , Volz A , Ludwig-Schwellinger E , Ebner T , Schaefer O |
| Ref : Drug Metabolism & Disposition: The Biological Fate of Chemicals , 42 :250 , 2014 |
| Abstract : Ishiguro_2014_Drug.Metab.Dispos_42_250 |
| ESTHER : Ishiguro_2014_Drug.Metab.Dispos_42_250 |
| PubMedSearch : Ishiguro_2014_Drug.Metab.Dispos_42_250 |
| PubMedID: 24212377 |
| Title : Identification of carboxylesterase-dependent dabigatran etexilate hydrolysis - Laizure_2014_Drug.Metab.Dispos_42_201 |
| Author(s) : Laizure SC , Parker RB , Herring VL , Hu ZY |
| Ref : Drug Metabolism & Disposition: The Biological Fate of Chemicals , 42 :201 , 2014 |
| Abstract : Laizure_2014_Drug.Metab.Dispos_42_201 |
| ESTHER : Laizure_2014_Drug.Metab.Dispos_42_201 |
| PubMedSearch : Laizure_2014_Drug.Metab.Dispos_42_201 |
| PubMedID: 24212379 |
| Gene_locus related to this paper: human-CES1 |