BHBT
General
Type : Terephthalate || Phthalate
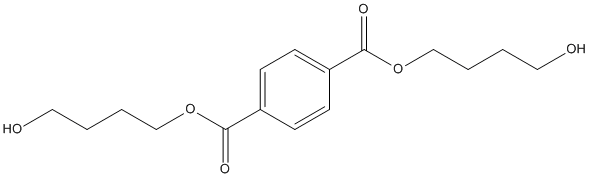
Chemical_Nomenclature : bis(4-hydroxybutyl) benzene-1,4-dicarboxylate
Canonical SMILES : C1=CC(=CC=C1C(=O)OCCCCO)C(=O)OCCCCO
InChI : InChI=1S\/C16H22O6\/c17-9-1-3-11-21-15(19)13-5-7-14(8-6-13)16(20)22-12-4-2-10-18\/h5-8,17-18H,1-4,9-12H2
InChIKey : MRLFFZIIRRKXBJ-UHFFFAOYSA-N
Other name(s) : bis(4-hydroxybutyl) terephthalate, bis(hydroxybutyl) terephthalate, Bis(4-hydroxybutyl) Benzene-1,4-dicarboxylate, 1,4-Benzenedicarboxylic acid, 1,4-bis(4-hydroxybutyl) ester, SCHEMBL128506, BTaB
Target
Families : Polyesterase-lipase-cutinase, Bacterial_lip_FamI.5
References (3)
| Title : Molecular and Biochemical Differences of the Tandem and Cold-Adapted PET Hydrolases Ple628 and Ple629, Isolated From a Marine Microbial Consortium - Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| Author(s) : Meyer-Cifuentes IE , Wu P , Zhao Y , Liu W , Neumann-Schaal M , Pfaff L , Barys J , Li Z , Gao J , Han X , Bornscheuer UT , Wei R , Ozturk B |
| Ref : Front Bioeng Biotechnol , 10 :930140 , 2022 |
| Abstract : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| ESTHER : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| PubMedSearch : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| PubMedID: 35935485 |
| Gene_locus related to this paper: 9zzzz-Ple628 , 9zzzz-Ple629 |
| Title : Substrate specificities of cutinases on aliphatic-aromatic polyesters and on their model substrates - Perz_2016_N.Biotechnol_33_295 |
| Author(s) : Perz V , Bleymaier K , Sinkel C , Kueper U , Bonnekessel M , Ribitsch D , Guebitz GM |
| Ref : N Biotechnol , 33 :295 , 2016 |
| Abstract : Perz_2016_N.Biotechnol_33_295 |
| ESTHER : Perz_2016_N.Biotechnol_33_295 |
| PubMedSearch : Perz_2016_N.Biotechnol_33_295 |
| PubMedID: 26594021 |
| Gene_locus related to this paper: humin-cut , thefu-q6a0i4 |
| Title : Hydrolysis of synthetic polyesters by Clostridium botulinum esterases - Perz_2016_Biotechnol.Bioeng_113_1024 |
| Author(s) : Perz V , Baumschlager A , Bleymaier K , Zitzenbacher S , Hromic A , Steinkellner G , Pairitsch A , Lyskowski A , Gruber K , Sinkel C , Kuper U , Ribitsch D , Guebitz GM |
| Ref : Biotechnol Bioeng , 113 :1024 , 2016 |
| Abstract : Perz_2016_Biotechnol.Bioeng_113_1024 |
| ESTHER : Perz_2016_Biotechnol.Bioeng_113_1024 |
| PubMedSearch : Perz_2016_Biotechnol.Bioeng_113_1024 |
| PubMedID: 26524601 |
| Gene_locus related to this paper: clobh-A5I3I2 , clobh-A51055 |