BETEB
General
Type : Terephthalate || Phthalate
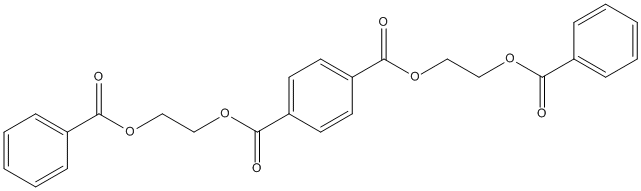
Chemical_Nomenclature : bis(2-benzoyloxyethyl) benzene-1,4-dicarboxylate
Canonical SMILES : C1=CC=C(C=C1)C(=O)OCCOC(=O)C2=CC=C(C=C2)C(=O)OCCOC(=O)C3=CC=CC=C3
InChI : InChI=1S\/C26H22O8\/c27-23(19-7-3-1-4-8-19)31-15-17-33-25(29)21-11-13-22(14-12-21)26(30)34-18-16-32-24(28)20-9-5-2-6-10-20\/h1-14H,15-18H2
InChIKey : JEWFVAIIRNKVIL-UHFFFAOYSA-N
Other name(s) : bis(2-(benzoyloxy)ethyl) terephthalate, bis(benzoyloxyethyl)terephthalate, 3PET, 3-PET, BaETaEBa, SCHEMBL547137, bis(benzoyloxyethyl) terephthalate, Terephthalic acid bis[2-(benzoyloxy)ethyl] ester, bis(2-benzoyloxyethyl) benzene-1,4-dicarboxylate
MW : 462.4
Formula : C26H22O8
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
References (14)
| Title : Structural Insights into (Tere)phthalate-Ester Hydrolysis by a Carboxylesterase and Its Role in Promoting PET Depolymerization - von Haugwitz_2022_ACS.Catal_12_15259 |
| Author(s) : von Haugwitz G , Han X , Pfaff L , Li Q , Wei H , Gao J , Methling K , Ao Y , Brack Y , Jan Mican J , Feiler CG , Weiss MS , Bednar D , Palm GJ , Lalk M , Lammers M , Damborsky J , Weber G , Liu W , Bornscheuer UT , Wei R |
| Ref : ACS Catal , 12 :15259 , 2022 |
| Abstract : von Haugwitz_2022_ACS.Catal_12_15259 |
| ESTHER : von Haugwitz_2022_ACS.Catal_12_15259 |
| PubMedSearch : von Haugwitz_2022_ACS.Catal_12_15259 |
| PubMedID: 36570084 |
| Gene_locus related to this paper: thefu-1831 |
| Title : Molecular and Biochemical Differences of the Tandem and Cold-Adapted PET Hydrolases Ple628 and Ple629, Isolated From a Marine Microbial Consortium - Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| Author(s) : Meyer-Cifuentes IE , Wu P , Zhao Y , Liu W , Neumann-Schaal M , Pfaff L , Barys J , Li Z , Gao J , Han X , Bornscheuer UT , Wei R , Ozturk B |
| Ref : Front Bioeng Biotechnol , 10 :930140 , 2022 |
| Abstract : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| ESTHER : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| PubMedSearch : Meyer-Cifuentes_2022_Front.Bioeng.Biotechnol_10_930140 |
| PubMedID: 35935485 |
| Gene_locus related to this paper: 9zzzz-Ple628 , 9zzzz-Ple629 |
| Title : Structure-function analysis of two closely related cutinases from Thermobifida cellulosilytica - Baath_2021_Biotechnol.Bioeng__ |
| Author(s) : Baath JA , Novy V , Carneiro LV , Guebitz GM , Olsson L , Westh P , Ribitsch D |
| Ref : Biotechnol Bioeng , : , 2021 |
| Abstract : Baath_2021_Biotechnol.Bioeng__ |
| ESTHER : Baath_2021_Biotechnol.Bioeng__ |
| PubMedSearch : Baath_2021_Biotechnol.Bioeng__ |
| PubMedID: 34755331 |
| Gene_locus related to this paper: thefu-q6a0i4 , thefu-q6a0i3 |
| Title : Current knowledge on enzymatic PET degradation and its possible application to waste stream management and other fields - Kawai_2019_Appl.Microbiol.Biotechnol_103_4253 |
| Author(s) : Kawai F , Kawabata T , Oda M |
| Ref : Applied Microbiology & Biotechnology , 103 :4253 , 2019 |
| Abstract : Kawai_2019_Appl.Microbiol.Biotechnol_103_4253 |
| ESTHER : Kawai_2019_Appl.Microbiol.Biotechnol_103_4253 |
| PubMedSearch : Kawai_2019_Appl.Microbiol.Biotechnol_103_4253 |
| PubMedID: 30957199 |
| Title : Substrate specificities of cutinases on aliphatic-aromatic polyesters and on their model substrates - Perz_2016_N.Biotechnol_33_295 |
| Author(s) : Perz V , Bleymaier K , Sinkel C , Kueper U , Bonnekessel M , Ribitsch D , Guebitz GM |
| Ref : N Biotechnol , 33 :295 , 2016 |
| Abstract : Perz_2016_N.Biotechnol_33_295 |
| ESTHER : Perz_2016_N.Biotechnol_33_295 |
| PubMedSearch : Perz_2016_N.Biotechnol_33_295 |
| PubMedID: 26594021 |
| Gene_locus related to this paper: humin-cut , thefu-q6a0i4 |
| Title : Hydrolysis of synthetic polyesters by Clostridium botulinum esterases - Perz_2016_Biotechnol.Bioeng_113_1024 |
| Author(s) : Perz V , Baumschlager A , Bleymaier K , Zitzenbacher S , Hromic A , Steinkellner G , Pairitsch A , Lyskowski A , Gruber K , Sinkel C , Kuper U , Ribitsch D , Guebitz GM |
| Ref : Biotechnol Bioeng , 113 :1024 , 2016 |
| Abstract : Perz_2016_Biotechnol.Bioeng_113_1024 |
| ESTHER : Perz_2016_Biotechnol.Bioeng_113_1024 |
| PubMedSearch : Perz_2016_Biotechnol.Bioeng_113_1024 |
| PubMedID: 26524601 |
| Gene_locus related to this paper: clobh-A5I3I2 , clobh-A51055 |
| Title : Surface engineering of a cutinase from Thermobifida cellulosilytica for improved polyester hydrolysis - Herrero_2013_Biotechnol.Bioeng_110_2581 |
| Author(s) : Herrero Acero E , Ribitsch D , Dellacher A , Zitzenbacher S , Marold A , Steinkellner G , Gruber K , Schwab H , Guebitz GM |
| Ref : Biotechnol Bioeng , 110 :2581 , 2013 |
| Abstract : Herrero_2013_Biotechnol.Bioeng_110_2581 |
| ESTHER : Herrero_2013_Biotechnol.Bioeng_110_2581 |
| PubMedSearch : Herrero_2013_Biotechnol.Bioeng_110_2581 |
| PubMedID: 23592055 |
| Title : Fusion of binding domains to Thermobifida cellulosilytica cutinase to tune sorption characteristics and enhancing PET hydrolysis - Ribitsch_2013_Biomacromolecules_14_1769 |
| Author(s) : Ribitsch D , Yebra AO , Zitzenbacher S , Wu J , Nowitsch S , Steinkellner G , Greimel K , Doliska A , Oberdorfer G , Gruber CC , Gruber K , Schwab H , Stana-Kleinschek K , Herrero Acero E , Guebitz GM |
| Ref : Biomacromolecules , 14 :1769 , 2013 |
| Abstract : Ribitsch_2013_Biomacromolecules_14_1769 |
| ESTHER : Ribitsch_2013_Biomacromolecules_14_1769 |
| PubMedSearch : Ribitsch_2013_Biomacromolecules_14_1769 |
| PubMedID: 23718548 |
| Gene_locus related to this paper: thefu-q6a0i4 |
| Title : A New Esterase from Thermobifida halotolerans Hydrolyses Polyethylene Terephthalate (PET) and Polylactic Acid (PLA) - Ribitsch_2012_Polymers_4_617 |
| Author(s) : Ribitsch D , Herrero Acero E , Greimel K , Dellacher A , Zitzenbacher S , Marold A , Rodriguez RD , Steinkellner G , Gruber K , Schwab H , Guebitz GM |
| Ref : Polymers , 4 :617 , 2012 |
| Abstract : Ribitsch_2012_Polymers_4_617 |
| ESTHER : Ribitsch_2012_Polymers_4_617 |
| PubMedSearch : Ribitsch_2012_Polymers_4_617 |
| PubMedID: |
| Gene_locus related to this paper: 9actn-h6wx58 |
| Title : Hydrolysis of polyethyleneterephthalate by p-nitrobenzylesterase from Bacillus subtilis - Ribitsch_2011_Biotechnol.Prog_27_951 |
| Author(s) : Ribitsch D , Heumann S , Trotscha E , Herrero Acero E , Greimel K , Leber R , Birner-Gruenberger R , Deller S , Eiteljoerg I , Remler P , Weber T , Siegert P , Maurer KH , Donelli I , Freddi G , Schwab H , Guebitz GM |
| Ref : Biotechnol Prog , 27 :951 , 2011 |
| Abstract : Ribitsch_2011_Biotechnol.Prog_27_951 |
| ESTHER : Ribitsch_2011_Biotechnol.Prog_27_951 |
| PubMedSearch : Ribitsch_2011_Biotechnol.Prog_27_951 |
| PubMedID: 21574267 |
| Gene_locus related to this paper: bacsu-pnbae |
| Title : Enzymes for the biofunctionalization of poly(ethylene terephthalate) - Zimmermann_2011_Adv.Biochem.Eng.Biotechnol_125_97 |
| Author(s) : Zimmermann W , Billig S |
| Ref : Adv Biochem Eng Biotechnol , 125 :97 , 2011 |
| Abstract : Zimmermann_2011_Adv.Biochem.Eng.Biotechnol_125_97 |
| ESTHER : Zimmermann_2011_Adv.Biochem.Eng.Biotechnol_125_97 |
| PubMedSearch : Zimmermann_2011_Adv.Biochem.Eng.Biotechnol_125_97 |
| PubMedID: 21076908 |
| Title : Enzymatic surface hydrolysis of poly(ethylene terephthalate) and bis(benzoyloxyethyl) terephthalate by lipase and cutinase in the presence of surface active molecules - Eberl_2009_J.Biotechnol_143_207 |
| Author(s) : Eberl A , Heumann S , Bruckner T , Araujo R , Cavaco-Paulo A , Kaufmann F , Kroutil W , Guebitz GM |
| Ref : J Biotechnol , 143 :207 , 2009 |
| Abstract : Eberl_2009_J.Biotechnol_143_207 |
| ESTHER : Eberl_2009_J.Biotechnol_143_207 |
| PubMedSearch : Eberl_2009_J.Biotechnol_143_207 |
| PubMedID: 19616594 |
| Title : New model substrates for enzymes hydrolysing polyethyleneterephthalate and polyamide fibres - Heumann_2006_J.Biochem.Biophys.Methods_69_89 |
| Author(s) : Heumann S , Eberl A , Pobeheim H , Liebminger S , Fischer-Colbrie G , Almansa E , Cavaco-Paulo A , Gubitz GM |
| Ref : Journal of Biochemical & Biophysical Methodsods , 69 :89 , 2006 |
| Abstract : Heumann_2006_J.Biochem.Biophys.Methods_69_89 |
| ESTHER : Heumann_2006_J.Biochem.Biophys.Methods_69_89 |
| PubMedSearch : Heumann_2006_J.Biochem.Biophys.Methods_69_89 |
| PubMedID: 16624419 |
| Title : Enzymatic degradation of poly(ethylene terephthalate): rapid hydrolyse using a hydrolase from T. fusca - Muller_2005_Macromol.Rapid.Commun_26_1400 |
| Author(s) : R.J. Muller, H. Schrader, J. Profe, K. Dresler, W.D. Deckwer |
| Ref : Macromol. Rapid Commun , 26 :1400 , 2005 |
| Abstract : Muller_2005_Macromol.Rapid.Commun_26_1400 |
| ESTHER : Muller_2005_Macromol.Rapid.Commun_26_1400 |
| PubMedSearch : Muller_2005_Macromol.Rapid.Commun_26_1400 |
| PubMedID: |
| Gene_locus related to this paper: thefu-1831 |