3'-Methyl-GR24
General
Type : Strigolactone || Strigolactone receptors ligand || Inden
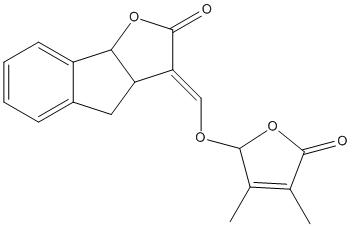
Chemical_Nomenclature : (3E)-3-[(3,4-dimethyl-5-oxo-2H-furan-2-yl)oxymethylidene]-4,8b-dihydro-3aH-indeno[1,2-b]furan-2-one
Canonical SMILES : CC1=C(C(=O)OC1OC=C2C3CC4=CC=CC=C4C3OC2=O)C
InChI : InChI=1S\/C18H16O5\/c1-9-10(2)18(23-16(9)19)21-8-14-13-7-11-5-3-4-6-12(11)15(13)22-17(14)20\/h3-6,8,13,15,18H,7H2,1-2H3\/b14-8+
InChIKey : ZDFFEMPWRSNISV-RIYZIHGNSA-N
Other name(s) : SCHEMBL19984943, SCHEMBL15726567, (+\/-)-3'-Methyl-GR24, Methyl-GR24, Methyl GR24, SCHEMBL14850471
MW : 312.3
Formula : C18H16O5
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
Families : RsbQ-like
References (3)
| Title : Expansion of the Strigolactone Profluorescent Probes Repertory: The Right Probe for the Right Application - de Saint Germain_2022_Front.Plant.Sci_13_887347 |
| Author(s) : de Saint Germain A , Clave G , Schouveiler P , Pillot JP , Singh AV , Chevalier A , Daignan Fornier S , Guillory A , Bonhomme S , Rameau C , Boyer FD |
| Ref : Front Plant Sci , 13 :887347 , 2022 |
| Abstract : de Saint Germain_2022_Front.Plant.Sci_13_887347 |
| ESTHER : de Saint Germain_2022_Front.Plant.Sci_13_887347 |
| PubMedSearch : de Saint Germain_2022_Front.Plant.Sci_13_887347 |
| PubMedID: 35720613 |
| Title : A Phelipanche ramosa KAI2 protein perceives strigolactones and isothiocyanates enzymatically - de Saint Germain_2021_Plant.Commun_2_100166 |
| Author(s) : de Saint Germain A , Jacobs A , Brun G , Pouvreau JB , Braem L , Cornu D , Clave G , Baudu E , Steinmetz V , Servajean V , Wicke S , Gevaert K , Simier P , Goormachtig S , Delavault P , Boyer FD |
| Ref : Plant Commun , 2 :100166 , 2021 |
| Abstract : de Saint Germain_2021_Plant.Commun_2_100166 |
| ESTHER : de Saint Germain_2021_Plant.Commun_2_100166 |
| PubMedSearch : de Saint Germain_2021_Plant.Commun_2_100166 |
| PubMedID: 34746757 |
| Gene_locus related to this paper: phera-PrKAI2c , phera-PrKAI2d1 , phera-PrKAI2d2 , phera-PrKAI2d3 , phera-PrKAI2d4 |
| Title : Structure-activity relationship studies of strigolactone-related molecules for branching inhibition in garden pea: molecule design for shoot branching - Boyer_2012_Plant.Physiol_159_1524 |
| Author(s) : Boyer FD , de Saint Germain A , Pillot JP , Pouvreau JB , Chen VX , Ramos S , Stevenin A , Simier P , Delavault P , Beau JM , Rameau C |
| Ref : Plant Physiol , 159 :1524 , 2012 |
| Abstract : Boyer_2012_Plant.Physiol_159_1524 |
| ESTHER : Boyer_2012_Plant.Physiol_159_1524 |
| PubMedSearch : Boyer_2012_Plant.Physiol_159_1524 |
| PubMedID: 22723084 |