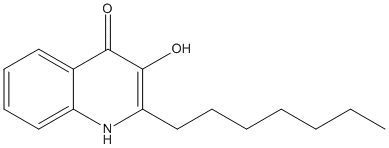
2-Heptyl-3-hydroxy-4-quinolone
General
Type : Quinoline || Natural
Chemical_Nomenclature : 2-heptyl-3-hydroxy-1H-quinolin-4-one
Canonical SMILES : CCCCCCCC1=C(C(=O)C2=CC=CC=C2N1)O
InChI : InChI=1S\/C16H21NO2\/c1-2-3-4-5-6-11-14-16(19)15(18)12-9-7-8-10-13(12)17-14\/h7-10,19H,2-6,11H2,1H3,(H,17,18)
InChIKey : CEIUIHOQDSVZJQ-UHFFFAOYSA-N
Other name(s) : 2-heptyl-3-hydroxy-4(1H)-quinolone, 2-Heptyl-3-hydroxy-quinolone, Pseudomonas quinolone signal, 2-heptyl-3-hydroxy-1H-quinolin-4-one, CHEMBL2426244, PQS
MW : 259.34
Formula : C16H21NO2
CAS_number :
PubChem :
UniChem :
Iuphar :
Target
Families : HOD-cofactorfree-dioxygenase
References (8)
| Title : Enzyme-Mediated Quenching of the Pseudomonas Quinolone Signal (PQS): A Comparison between Naturally Occurring and Engineered PQS-Cleaving Dioxygenases - Arranz San Martin_2022_Biomolecules_12_170 |
| Author(s) : Arranz San Martin A , Vogel J , Wullich SC , Quax WJ , Fetzner S |
| Ref : Biomolecules , 12 :170 , 2021 |
| Abstract : Arranz San Martin_2022_Biomolecules_12_170 |
| ESTHER : Arranz San Martin_2022_Biomolecules_12_170 |
| PubMedSearch : Arranz San Martin_2022_Biomolecules_12_170 |
| PubMedID: |
| Gene_locus related to this paper: artsp-hod , strbb-d7bw96 , mycab-x8en65 , nocfa-q5yp20 |
| Title : Structural basis for recognition and ring-cleavage of the Pseudomonas quinolone signal (PQS) by AqdC, a mycobacterial dioxygenase of the alpha\/beta-hydrolase fold family - Wullich_2019_J.Struct.Biol_207_287 |
| Author(s) : Wullich SC , Kobus S , Wienhold M , Hennecke U , Smits SHJ , Fetzner S |
| Ref : J Struct Biol , 207 :287 , 2019 |
| Abstract : Wullich_2019_J.Struct.Biol_207_287 |
| ESTHER : Wullich_2019_J.Struct.Biol_207_287 |
| PubMedSearch : Wullich_2019_J.Struct.Biol_207_287 |
| PubMedID: 31228546 |
| Gene_locus related to this paper: mycab-x8en65 |
| Title : Mycobacterium abscessus subsp. abscessus Is Capable of Degrading Pseudomonas aeruginosa Quinolone Signals - Birmes_2017_Front.Microbiol_8_339 |
| Author(s) : Birmes FS , Wolf T , Kohl TA , Ruger K , Bange F , Kalinowski J , Fetzner S |
| Ref : Front Microbiol , 8 :339 , 2017 |
| Abstract : Birmes_2017_Front.Microbiol_8_339 |
| ESTHER : Birmes_2017_Front.Microbiol_8_339 |
| PubMedSearch : Birmes_2017_Front.Microbiol_8_339 |
| PubMedID: 28303132 |
| Gene_locus related to this paper: rhoer-aqdC1 , rhoer-aqdC2 |
| Title : Enzyme-Mediated Quenching of the Pseudomonas Quinolone Signal (PQS) Promotes Biofilm Formation of Pseudomonas aeruginosa by Increasing Iron Availability - Tettmann_2016_Front.Microbiol_7_1978 |
| Author(s) : Tettmann B , Niewerth C , Kirschhofer F , Neidig A , Dotsch A , Brenner-Weiss G , Fetzner S , Overhage J |
| Ref : Front Microbiol , 7 :1978 , 2016 |
| Abstract : Tettmann_2016_Front.Microbiol_7_1978 |
| ESTHER : Tettmann_2016_Front.Microbiol_7_1978 |
| PubMedSearch : Tettmann_2016_Front.Microbiol_7_1978 |
| PubMedID: 28018312 |
| Title : Rhodococcus erythropolis BG43 Genes Mediating Pseudomonas aeruginosa Quinolone Signal Degradation and Virulence Factor Attenuation - Muller_2015_Appl.Environ.Microbiol_81_7720 |
| Author(s) : Muller C , Birmes FS , Ruckert C , Kalinowski J , Fetzner S |
| Ref : Applied Environmental Microbiology , 81 :7720 , 2015 |
| Abstract : Muller_2015_Appl.Environ.Microbiol_81_7720 |
| ESTHER : Muller_2015_Appl.Environ.Microbiol_81_7720 |
| PubMedSearch : Muller_2015_Appl.Environ.Microbiol_81_7720 |
| PubMedID: 26319870 |
| Gene_locus related to this paper: rhoer-aqdC1 , rhoer-aqdC2 , rhoer-aqda1 |
| Title : Conversion of the Pseudomonas aeruginosa Quinolone Signal and Related Alkylhydroxyquinolines by Rhodococcus sp. Strain BG43 - Muller_2014_Appl.Environ.Microbiol_80_7266 |
| Author(s) : Muller C , Birmes FS , Niewerth H , Fetzner S |
| Ref : Applied Environmental Microbiology , 80 :7266 , 2014 |
| Abstract : Muller_2014_Appl.Environ.Microbiol_80_7266 |
| ESTHER : Muller_2014_Appl.Environ.Microbiol_80_7266 |
| PubMedSearch : Muller_2014_Appl.Environ.Microbiol_80_7266 |
| PubMedID: 25239889 |
| Gene_locus related to this paper: rhoer-aqdC1 , rhoer-aqdC2 |
| Title : Validation of PqsD as an anti-biofilm target in Pseudomonas aeruginosa by development of small-molecule inhibitors - Storz_2012_J.Am.Chem.Soc_134_16143 |
| Author(s) : Storz MP , Maurer CK , Zimmer C , Wagner N , Brengel C , de Jong JC , Lucas S , Musken M , Haussler S , Steinbach A , Hartmann RW |
| Ref : Journal of the American Chemical Society , 134 :16143 , 2012 |
| Abstract : Storz_2012_J.Am.Chem.Soc_134_16143 |
| ESTHER : Storz_2012_J.Am.Chem.Soc_134_16143 |
| PubMedSearch : Storz_2012_J.Am.Chem.Soc_134_16143 |
| PubMedID: 22992202 |
| Title : Quorum sensing by 2-alkyl-4-quinolones in Pseudomonas aeruginosa and other bacterial species - Dubern_2008_Mol.Biosyst_4_882 |
| Author(s) : Dubern JF , Diggle SP |
| Ref : Mol Biosyst , 4 :882 , 2008 |
| Abstract : Dubern_2008_Mol.Biosyst_4_882 |
| ESTHER : Dubern_2008_Mol.Biosyst_4_882 |
| PubMedSearch : Dubern_2008_Mol.Biosyst_4_882 |
| PubMedID: 18704225 |