WAY-121898
IC50 650nM pig-i3ltk9 (Homan et al. 1997) Commons, T.J., LaClair, C.M., Strike, D.P., Clark, D.E., McKean, M.L., and Adelman, S.J. (1992) Am. Chem. Soc., 203 Meeting, MEDI 139 (Abstract)- McKean, M.L., Commons, T.J., Berens, M.S., Hsu, P.L., Ackerman, D.M., Steiner, K.E., and Adelman, S.J. (1992) FASEB J. 6, A1388 (Abstract) - Mewshaw, R.E., Commons, T.J., and Strike, D.P. (1991) European Patent Number EP0428385 A - Adelman, S.J., Commons, T.J., McKean, M.L., Day, C.E., and Steiner, K.E. (1992) XI Int. Symposium on Drugs Affecting Lipid Metabolism 87 (Abstract) - Gallo, L.L., Srivastava, S., Satchithanandam, S., Steiner, K.E., McKean, M.L., and Adelman, S.J. (1992) FASEB J. 6, A1388 (Abstract).
General
Type : Carbamate,Piperidine,Phenoxyphenyl
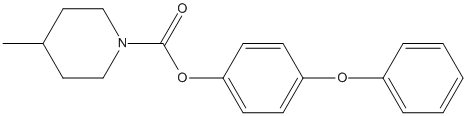
Chemical_Nomenclature : (4-phenoxyphenyl) 4-methylpiperidine-1-carboxylate
Canonical SMILES : CC1CCN(CC1)C(=O)OC2=CC=C(C=C2)OC3=CC=CC=C3
InChI : InChI=1S\/C19H21NO3\/c1-15-11-13-20(14-12-15)19(21)23-18-9-7-17(8-10-18)22-16-5-3-2-4-6-16\/h2-10,15H,11-14H2,1H3
InChIKey : QRGQONAZHIKMGC-UHFFFAOYSA-N
Other name(s) : WAY-121,898,1-Piperidinecarboxylic acid, 4-methyl-, 4-phenoxyphenyl ester,ACMC-20mw0l,CHEMBL267052,SCHEMBL2597837,ZINC14493
MW : 311.4
Formula : C19H21NO3
CAS_number : 136100-14-6
PubChem : 9796754
UniChem : QRGQONAZHIKMGC-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : WAY-121898 ligand of proteins in family: Cholesterol_esterase
Stucture :
Protein : pig-i3ltk9
References (3)
| Title : The biology and chemistry of hyperlipidemia - Jain_2007_Bioorg.Med.Chem_15_4674 |
| Author(s) : Jain KS , Kathiravan MK , Somani RS , Shishoo CJ |
| Ref : Bioorganic & Medicinal Chemistry , 15 :4674 , 2007 |
| Abstract : Jain_2007_Bioorg.Med.Chem_15_4674 |
| ESTHER : Jain_2007_Bioorg.Med.Chem_15_4674 |
| PubMedSearch : Jain_2007_Bioorg.Med.Chem_15_4674 |
| PubMedID: 17521912 |
| Title : Lipid-lowering effects of WAY-121,898, an inhibitor of pancreatic cholesteryl ester hydrolase - Krause_1998_Lipids_33_489 |
| Author(s) : Krause BR , Sliskovic DR , Anderson M , Homan R |
| Ref : Lipids , 33 :489 , 1998 |
| Abstract : Krause_1998_Lipids_33_489 |
| ESTHER : Krause_1998_Lipids_33_489 |
| PubMedSearch : Krause_1998_Lipids_33_489 |
| PubMedID: 9625596 |
| Gene_locus related to this paper: human-CEL , ratno-balip , mesau-a0a1u8cxs4 |
| Title : Established and Emerging Strategies for Inhibition of Cholesterol Absorption - Homan_1997_Curr.Pharma.Design_3_29 |
| Author(s) : Homan R , Krause BR |
| Ref : Curr.Pharma.Design , 3 :29 , 1997 |
| Abstract : Homan_1997_Curr.Pharma.Design_3_29 |
| ESTHER : Homan_1997_Curr.Pharma.Design_3_29 |
| PubMedSearch : Homan_1997_Curr.Pharma.Design_3_29 |
| PubMedID: |
| Gene_locus related to this paper: human-CEL , ratno-balip , mesau-a0a1u8cxs4 |