Ro-2-0683
Specific inhibitor of BChE, used for phenotyping K and Atypical variants. Entry of reference human-BCHE
General
Type : Trimethylammonium,Carbamate
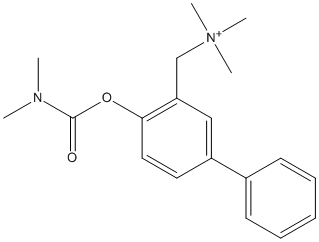
Chemical_Nomenclature : [2-(dimethylcarbamoyloxy)-5-phenylphenyl]methyl-trimethylazanium\;bromide
Canonical SMILES : CN(C)C(=O)OC1=C(C=C(C=C1)C2=CC=CC=C2)C[N+](C)(C)C.[Br-]
InChI : InChI=1S\/C19H25N2O2.BrH\/c1-20(2)19(22)23-18-12-11-16(15-9-7-6-8-10-15)13-17(18)14-21(3,4)5\;\/h6-13H,14H2,1-5H3\;1H\/q+1\;\/p-1
InChIKey : BKQQATFNHNQNJL-UHFFFAOYSA-M
Other name(s) : RO2-0683,RO 20683,Ro-02-0683,dimethyl carbamate of (2-hydroxy-5-phenylbenzyl)-trimethyl-ammonium bromide,Nu-683,NSC 306692,Ro 2-0683,(2-Hydroxy-5-phenylbenzyl)trimethylammonium bromide
MW : 393.318
Formula : C19H25BrN2O2
CAS_number : 1954-17-2
PubChem : 164903
UniChem : BKQQATFNHNQNJL-UHFFFAOYSA-M
IUPHAR :
Wikipedia :
Target
References (19)
| Title : An improvement in segregation of human butyrylcholinesterase phenotypes having the fluoride-resistant variants - Kovarik_2003_Arh.Hig.Rada.Toksikol_54_239 |
| Author(s) : Kovarik Z , Simeon-Rudolf V |
| Ref : Arh Hig Rada Toksikol , 54 :239 , 2003 |
| Abstract : Kovarik_2003_Arh.Hig.Rada.Toksikol_54_239 |
| ESTHER : Kovarik_2003_Arh.Hig.Rada.Toksikol_54_239 |
| PubMedSearch : Kovarik_2003_Arh.Hig.Rada.Toksikol_54_239 |
| PubMedID: 14994645 |
| Title : Adamantyl tenocyclidines--adjuvant therapy in poisoning with organophosphorus compounds and carbamates - Skare_2002_Arch.Toxicol_76_173 |
| Author(s) : Skare D , Radic B , Lucic A , Peraica M , Domijan AM , Milkovic-Kraus S , Bradamante V , Jukic I |
| Ref : Archives of Toxicology , 76 :173 , 2002 |
| Abstract : Skare_2002_Arch.Toxicol_76_173 |
| ESTHER : Skare_2002_Arch.Toxicol_76_173 |
| PubMedSearch : Skare_2002_Arch.Toxicol_76_173 |
| PubMedID: 11967623 |
| Title : Warning card as a prevention of prolonged apnea in children - Lajtman_2002_Coll.Antropol_26_129 |
| Author(s) : Lajtman Z , Surina B , Bartolek D , Car D , Gasparovic S , Kirincic N , Rudez J |
| Ref : Coll Antropol , 26 :129 , 2002 |
| Abstract : Lajtman_2002_Coll.Antropol_26_129 |
| ESTHER : Lajtman_2002_Coll.Antropol_26_129 |
| PubMedSearch : Lajtman_2002_Coll.Antropol_26_129 |
| PubMedID: 12674844 |
| Title : An explanation for the different inhibitory characteristics of human serum butyrylcholinesterase phenotypes deriving from inhibition of atypical heterozygotes - Simeon-Rudolf_1999_Chem.Biol.Interact_119-120_159 |
| Author(s) : Simeon-Rudolf V , Kovarik Z , Skrinjaric-Spoljar M , Evans RT |
| Ref : Chemico-Biological Interactions , 119-120 :159 , 1999 |
| Abstract : Simeon-Rudolf_1999_Chem.Biol.Interact_119-120_159 |
| ESTHER : Simeon-Rudolf_1999_Chem.Biol.Interact_119-120_159 |
| PubMedSearch : Simeon-Rudolf_1999_Chem.Biol.Interact_119-120_159 |
| PubMedID: 10421449 |
| Title : Amino acid residues involved in the interaction of acetylcholinesterase and butyrylcholinesterase with the carbamates Ro 02-0683 and bambuterol, and with terbutaline - Kovarik_1999_Biochim.Biophys.Acta_1433_261 |
| Author(s) : Kovarik Z , Radic Z , Grgas B , Skrinjaric-Spoljar M , Reiner E , Simeon-Rudolf V |
| Ref : Biochimica & Biophysica Acta , 1433 :261 , 1999 |
| Abstract : Kovarik_1999_Biochim.Biophys.Acta_1433_261 |
| ESTHER : Kovarik_1999_Biochim.Biophys.Acta_1433_261 |
| PubMedSearch : Kovarik_1999_Biochim.Biophys.Acta_1433_261 |
| PubMedID: 10446376 |
| Title : Identification of human plasma cholinesterase variants in 6,688 individuals using biochemical analysis - Jensen_1995_Acta.Anaesthesiol.Scand_39_157 |
| Author(s) : Jensen FS , Skovgaard LT , Viby-Mogensen J |
| Ref : Acta Anaesthesiologica Scandinavica , 39 :157 , 1995 |
| Abstract : Jensen_1995_Acta.Anaesthesiol.Scand_39_157 |
| ESTHER : Jensen_1995_Acta.Anaesthesiol.Scand_39_157 |
| PubMedSearch : Jensen_1995_Acta.Anaesthesiol.Scand_39_157 |
| PubMedID: 7793180 |
| Title : An improved method for butyrylcholinesterase phenotyping [published erratum appears in Biochem Genet 1994 Oct\;32(9-10):379] - Picheth_1994_Biochem.Genet_32_83 |
| Author(s) : Picheth G , Fadel-Picheth C , Primo-Parmo SL , Chautard-Freire-Maia EA , Vieira MM |
| Ref : Biochemical Genetics , 32 :83 , 1994 |
| Abstract : Picheth_1994_Biochem.Genet_32_83 |
| ESTHER : Picheth_1994_Biochem.Genet_32_83 |
| PubMedSearch : Picheth_1994_Biochem.Genet_32_83 |
| PubMedID: 7980387 |
| Title : Plasma cholinesterase: gene and variations - Pantuck_1993_Anesth.Analg_77_380 |
| Author(s) : Pantuck EJ |
| Ref : Anesthesia & Analgesia , 77 :380 , 1993 |
| Abstract : Pantuck_1993_Anesth.Analg_77_380 |
| ESTHER : Pantuck_1993_Anesth.Analg_77_380 |
| PubMedSearch : Pantuck_1993_Anesth.Analg_77_380 |
| PubMedID: 8346840 |
| Title : Kinetics of the inhibition of human serum cholinesterase phenotypes with the dimethylcarbamate of (2-hydroxy-5-phenylbenzyl)-trimethylammonium bromide (Ro 02-0683) - Prester_1991_Biochem.Pharmacol_42_2313 |
| Author(s) : Prester L , Simeon V |
| Ref : Biochemical Pharmacology , 42 :2313 , 1991 |
| Abstract : Prester_1991_Biochem.Pharmacol_42_2313 |
| ESTHER : Prester_1991_Biochem.Pharmacol_42_2313 |
| PubMedSearch : Prester_1991_Biochem.Pharmacol_42_2313 |
| PubMedID: 1764116 |
| Title : CHE1 UF serum cholinesterase phenotype in whites and non-whites from southern Brazil as determined by a new method - Alcantara_1991_Hum.Hered_41_103 |
| Author(s) : Alcantara VM , Chautard-Freire-Maia EA , Culpi L |
| Ref : Hum Hered , 41 :103 , 1991 |
| Abstract : Alcantara_1991_Hum.Hered_41_103 |
| ESTHER : Alcantara_1991_Hum.Hered_41_103 |
| PubMedSearch : Alcantara_1991_Hum.Hered_41_103 |
| PubMedID: 1855781 |
| Title : Genetic variants of human serum cholinesterase influence metabolism of the muscle relaxant succinylcholine. - Lockridge_1990_Pharmacol.Ther_47_35 |
| Author(s) : Lockridge O |
| Ref : Pharmacol Ther , 47 :35 , 1990 |
| Abstract : Lockridge_1990_Pharmacol.Ther_47_35 |
| ESTHER : Lockridge_1990_Pharmacol.Ther_47_35 |
| PubMedSearch : Lockridge_1990_Pharmacol.Ther_47_35 |
| PubMedID: 2195556 |
| Gene_locus related to this paper: human-BCHE |
| Title : Recognition of the Ek1Ek1 homozygote for plasma cholinesterase - Whittaker_1990_Hum.Hered_40_247 |
| Author(s) : Whittaker M , Britten JJ |
| Ref : Hum Hered , 40 :247 , 1990 |
| Abstract : Whittaker_1990_Hum.Hered_40_247 |
| ESTHER : Whittaker_1990_Hum.Hered_40_247 |
| PubMedSearch : Whittaker_1990_Hum.Hered_40_247 |
| PubMedID: 2379931 |
| Title : Differential inhibition of plasma cholinesterase variants using the dibutyrate analogue of pancuronium bromide - Whittaker_1981_Hum.Hered_31_242 |
| Author(s) : Whittaker M , Britten JJ |
| Ref : Hum Hered , 31 :242 , 1981 |
| Abstract : Whittaker_1981_Hum.Hered_31_242 |
| ESTHER : Whittaker_1981_Hum.Hered_31_242 |
| PubMedSearch : Whittaker_1981_Hum.Hered_31_242 |
| PubMedID: 7287015 |
| Title : Cholinergic effects on bull and chimpanzee sperm motility - |
| Author(s) : McGrady AV , Nelson L |
| Ref : Biol Reprod , 15 :248 , 1976 |
| PubMedID: 963150 |
| Title : The effects of anticholinesterases on synaptic transmission through nicotinic and muscarinic receptors in rat sympathetic ganglia in vivo - Drew_1974_Br.J.Pharmacol_52_51 |
| Author(s) : Drew GM , Leach GD |
| Ref : British Journal of Pharmacology , 52 :51 , 1974 |
| Abstract : Drew_1974_Br.J.Pharmacol_52_51 |
| ESTHER : Drew_1974_Br.J.Pharmacol_52_51 |
| PubMedSearch : Drew_1974_Br.J.Pharmacol_52_51 |
| PubMedID: 4375534 |
| Title : Action decurarisante du bromure de dimethylcarbamate de (2-hydroxy-5-phenylbenzyl)-trimethylammonium (Nu-683) - |
| Author(s) : Mazella H , Migliaro E |
| Ref : Archives Internationales de Pharmacodynamie et de Therapie , 79 :362 , 1949 |
| PubMedID: 18150729 |
| Title : Tubocurarine antagonism and inhibition of cholinesterases - |
| Author(s) : Blaschko H , Bulbring E , Chou TC |
| Ref : British Journal of Pharmacology , 4 :29 , 1949 |
| PubMedID: 18113150 |
| Title : Studies on cholinesterase: 5. The selective inhibition of pseudo-cholinesterase in vivo - |
| Author(s) : Hawkins RD , Gunter JM |
| Ref : Biochemical Journal , 40 :192 , 1946 |
| PubMedID: 16747984 |