Prusogliptin
General
Type : Gliptin,Trifluoro,Cyanide,Pyrrolidine
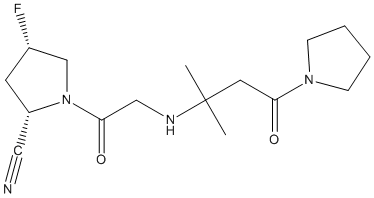
Chemical_Nomenclature : (2S,4S)-4-fluoro-1-[2-[(2-methyl-4-oxo-4-pyrrolidin-1-ylbutan-2-yl)amino]acetyl]pyrrolidine-2-carbonitrile
Canonical SMILES : CC(C)(CC(=O)N1CCCC1)NCC(=O)N2CC(CC2C#N)F
InChI : InChI=1S\/C16H25FN4O2\/c1-16(2,8-14(22)20-5-3-4-6-20)19-10-15(23)21-11-12(17)7-13(21)9-18\/h12-13,19H,3-8,10-11H2,1-2H3\/t12-,13-\/m0\/s1
InChIKey : VQKSCYBKUIDZEI-STQMWFEESA-N
Other name(s) : DBPR-108,DBPR108,UNII-E329HG23ZT,E329HG23ZT,CHEMBL1082462,SCHEMBL2401277,BDBM50320120,ZINC43207516
MW : 324.39
Formula : C16H25FN4O2
CAS_number : 1186426-66-3
PubChem : 44201003
UniChem : VQKSCYBKUIDZEI-STQMWFEESA-N
IUPHAR :
Wikipedia :
Target
Families : Prusogliptin ligand of proteins in family: DPP4N_Peptidase_S9
Stucture :
Protein : human-DPP4
References (5)
| Title : Combination of retagliptin and henagliflozin as add-on therapy to metformin in patients with type 2 diabetes inadequately controlled with metformin: A multicentre, randomized, double-blind, active-controlled, phase 3 trial - Wang_2024_Diabetes.Obes.Metab__ |
| Author(s) : Wang W , Guo X , Zhang C , Ning T , Ma G , Huang Y , Jia R , Zhou D , Cao M , Zhang T , Yao L , Yuan J , Chen L , Wang Y , Jiang C , Dong X , Chen M , Gu Q , Zhang L , Fu Y , Pan T , Bi Y , Song W , Xu J , Lu W , Sun X , Ye Z , Zhang D , Peng L , Lin X , Dai W , Wang Q , Yang W |
| Ref : Diabetes Obes Metab , : , 2024 |
| Abstract : Wang_2024_Diabetes.Obes.Metab__ |
| ESTHER : Wang_2024_Diabetes.Obes.Metab__ |
| PubMedSearch : Wang_2024_Diabetes.Obes.Metab__ |
| PubMedID: 38221859 || 38618970 |
| Title : Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial - Xu_2022_Diabetes.Obes.Metab__ |
| Author(s) : Xu J , Ling H , Geng J , Huang Y , Xie Y , Zheng H , Niu H , Zhang T , Yuan J , Xiao X |
| Ref : Diabetes Obes Metab , : , 2022 |
| Abstract : Xu_2022_Diabetes.Obes.Metab__ |
| ESTHER : Xu_2022_Diabetes.Obes.Metab__ |
| PubMedSearch : Xu_2022_Diabetes.Obes.Metab__ |
| PubMedID: 35791646 |
| Title : DBPR108, a novel dipeptidyl peptidase-4 inhibitor with antihyperglycemic activity - Yeh_2021_Life.Sci_278_119574 |
| Author(s) : Yeh KC , Yeh TK , Huang CY , Hu CB , Wang MH , Huang YW , Chou LH , Ho HH , Song JS , Hsu T , Jiaang WT , Chao YS , Chen CT |
| Ref : Life Sciences , 278 :119574 , 2021 |
| Abstract : Yeh_2021_Life.Sci_278_119574 |
| ESTHER : Yeh_2021_Life.Sci_278_119574 |
| PubMedSearch : Yeh_2021_Life.Sci_278_119574 |
| PubMedID: 33961850 |
| Title : Efficacy and safety of DBPR108 monotherapy in patients with type 2 diabetes: a 12-week, randomized, double-blind, placebo-controlled, phase II clinical trial - Wang_2020_Curr.Med.Res.Opin_36_1107 |
| Author(s) : Wang W , Yao J , Guo X , Guo Y , Yan C , Liu K , Zhang Y , Wang X , Li H , Wen Z , Li S , Xiao X , Liu W , Li Z , Zhang L , Shao S , Ye S , Qin G , Li Y , Li F , Zhang X , Li X , Peng Y , Deng H , Xu X , Zhou L , Huang Y , Cao M , Xia X , Shi M , Dou J , Yuan J |
| Ref : Curr Med Res Opin , 36 :1107 , 2020 |
| Abstract : Wang_2020_Curr.Med.Res.Opin_36_1107 |
| ESTHER : Wang_2020_Curr.Med.Res.Opin_36_1107 |
| PubMedSearch : Wang_2020_Curr.Med.Res.Opin_36_1107 |
| PubMedID: 32338063 |
| Title : (2S,4S)-1-[2-(1,1-dimethyl-3-oxo-3-pyrrolidin-1-yl-propylamino)acetyl]-4-fluoro-p yrrolidine-2-carbonitrile: a potent, selective, and orally bioavailable dipeptide-derived inhibitor of dipeptidyl peptidase IV - Yeh_2010_Bioorg.Med.Chem.Lett_20_3596 |
| Author(s) : Yeh TK , Tsai TY , Hsu T , Cheng JH , Chen X , Song JS , Shy HS , Chiou MC , Chien CH , Tseng YJ , Huang CY , Yeh KC , Huang YL , Huang CH , Huang YW , Wang MH , Tang HK , Chao YS , Chen CT , Jiaang WT |
| Ref : Bioorganic & Medicinal Chemistry Lett , 20 :3596 , 2010 |
| Abstract : Yeh_2010_Bioorg.Med.Chem.Lett_20_3596 |
| ESTHER : Yeh_2010_Bioorg.Med.Chem.Lett_20_3596 |
| PubMedSearch : Yeh_2010_Bioorg.Med.Chem.Lett_20_3596 |
| PubMedID: 20483603 |