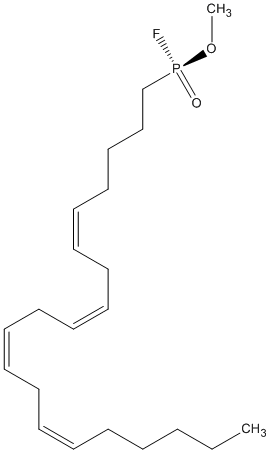
Methyl-arachidonyl-fluorophosphonate
found also in complex with patatin-17 and fatty acid amide hydrolase (amidase superfamily) || (MAFP) and related analogs have been shown to inhibit fatty acid amidohydrolase activity (FAAH)(not an alpha/beta hydrolase), the enzyme responsible for hydrolysis of the endogenous cannabinoid ligand anandamide. MAFP inhibits ABHD4. and PLA2G15 which are alpha/beta hydrolases (and also GIVA PLA2 not ABHyd)
General
Type : Cannabinoid-Receptor-ligand,Organophosphate,FAAH inhibitor,Multitarget
Chemical_Nomenclature : (5Z,8Z,11Z,14Z)-1-[fluoro(methoxy)phosphoryl]icosa-5,8,11,14-tetraene
Canonical SMILES : CCCCCC=CCC=CCC=CCC=CCCCCP(=O)(OC)F
InChI : InChI=1S\/C21H36FO2P\/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-25(22,23)24-2\/h7-8,10-11,13-14,16-17H,3-6,9,12,15,18-21H2,1-2H3\/b8-7-,11-10-,14-13-,17-16-
InChIKey : KWKZCGMJGHHOKJ-ZKWNWVNESA-N
Other name(s) : MAFP,Methyl arachidonyl fluorophosphonate,CHEMBL113262,methyl (S)-(5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraen-1-ylphosphonofluoridate
MW : 370.48
Formula : C21H36FO2P
CAS_number : 188404-10-6
PubChem : 10429254
UniChem : KWKZCGMJGHHOKJ-ZKWNWVNESA-N
IUPHAR :
Wikipedia :
Target
Families : Methyl-arachidonyl-fluorophosphonate ligand of proteins in family: CGI-58_ABHD5_ABHD4 || PC-sterol_acyltransferase || Monoglyceridelipase_lysophospholip
Stucture :
References (11)
| Title : Structure and Dynamics of an Archeal Monoglyceride Lipase from Palaeococcus ferrophilus as Revealed by Crystallography and In Silico Analysis - Labar_2021_Biomolecules_11_533 |
| Author(s) : Labar G , Brandt N , Flaba A , Wouters J , Leherte L |
| Ref : Biomolecules , 11 :533 , 2021 |
| Abstract : Labar_2021_Biomolecules_11_533 |
| ESTHER : Labar_2021_Biomolecules_11_533 |
| PubMedSearch : Labar_2021_Biomolecules_11_533 |
| PubMedID: 33916727 |
| Gene_locus related to this paper: 9eury-6QE2 |
| Title : An optimized spectrophotometric assay reveals increased activity of enzymes involved in 2-arachidonoyl glycerol turnover in the cerebral cortex of a rat model of Alzheimer's disease - Rodrigues_2020_Eur.J.Neurosci__ |
| Author(s) : Rodrigues MS , Ferreira C , Dias C , Pliassova A , Souza L , Ledo A , Laranjinha J , Cunha RA , Kofalvi A |
| Ref : European Journal of Neuroscience , : , 2020 |
| Abstract : Rodrigues_2020_Eur.J.Neurosci__ |
| ESTHER : Rodrigues_2020_Eur.J.Neurosci__ |
| PubMedSearch : Rodrigues_2020_Eur.J.Neurosci__ |
| PubMedID: 32813905 |
| Title : Human leukocytes differentially express endocannabinoid-glycerol lipases and hydrolyze 2-arachidonoyl-glycerol and its metabolites from the 15-lipoxygenase and cyclooxygenase pathways - Turcotte_2019_J.Leukoc.Biol_106_1337 |
| Author(s) : Turcotte C , Dumais E , Archambault AS , Martin C , Blanchet MR , Bissonnette E , Boulet LP , Laviolette M , Di Marzo V , Flamand N |
| Ref : J Leukoc Biol , 106 :1337 , 2019 |
| Abstract : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| ESTHER : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| PubMedSearch : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| PubMedID: 31556464 |
| Gene_locus related to this paper: human-ABHD6 , human-ABHD12 , human-ABHD16A , human-CES1 , human-LYPLA2 , human-PPT1 |
| Title : Ghrelin Octanoylation Is Completely Stabilized in Biological Samples by Alkyl Fluorophosphonates - McGovern-Gooch_2016_Endocrinology_157_4330 |
| Author(s) : McGovern-Gooch KR , Rodrigues T , Darling JE , Sieburg MA , Abizaid A , Hougland JL |
| Ref : Endocrinology , 157 :4330 , 2016 |
| Abstract : McGovern-Gooch_2016_Endocrinology_157_4330 |
| ESTHER : McGovern-Gooch_2016_Endocrinology_157_4330 |
| PubMedSearch : McGovern-Gooch_2016_Endocrinology_157_4330 |
| PubMedID: 27623288 |
| Title : Structure and function of lysosomal phospholipase A2 and lecithin:cholesterol acyltransferase - Glukhova_2015_Nat.Commun_6_6250 |
| Author(s) : Glukhova A , Hinkovska-Galcheva V , Kelly R , Abe A , Shayman JA , Tesmer JJ |
| Ref : Nat Commun , 6 :6250 , 2015 |
| Abstract : Glukhova_2015_Nat.Commun_6_6250 |
| ESTHER : Glukhova_2015_Nat.Commun_6_6250 |
| PubMedSearch : Glukhova_2015_Nat.Commun_6_6250 |
| PubMedID: 25727495 |
| Gene_locus related to this paper: human-LCAT |
| Title : Phospholipase A\/Acyltransferase enzyme activity of H-rev107 inhibits the H-RAS signaling pathway - Wang_2014_J.Biomed.Sci_21_36 |
| Author(s) : Wang CH , Shyu RY , Wu CC , Tsai TC , Wang LK , Chen ML , Jiang SY , Tsai FM |
| Ref : J Biomed Sci , 21 :36 , 2014 |
| Abstract : Wang_2014_J.Biomed.Sci_21_36 |
| ESTHER : Wang_2014_J.Biomed.Sci_21_36 |
| PubMedSearch : Wang_2014_J.Biomed.Sci_21_36 |
| PubMedID: 24884338 |
| Title : Association between Pseudomonas aeruginosa type III secretion, antibiotic resistance, and clinical outcome: a review - Sawa_2014_Crit.Care_18_668 |
| Author(s) : Sawa T , Shimizu M , Moriyama K , Wiener-Kronish JP |
| Ref : Crit Care , 18 :668 , 2014 |
| Abstract : Sawa_2014_Crit.Care_18_668 |
| ESTHER : Sawa_2014_Crit.Care_18_668 |
| PubMedSearch : Sawa_2014_Crit.Care_18_668 |
| PubMedID: 25672496 |
| Title : Inhibition of phospholipase A1, lipase and galactolipase activities of pancreatic lipase-related protein 2 by methyl arachidonyl fluorophosphonate (MAFP) - Amara_2012_Biochim.Biophys.Acta_1821_1379 |
| Author(s) : Amara S , Delorme V , Record M , Carriere F |
| Ref : Biochimica & Biophysica Acta , 1821 :1379 , 2012 |
| Abstract : Amara_2012_Biochim.Biophys.Acta_1821_1379 |
| ESTHER : Amara_2012_Biochim.Biophys.Acta_1821_1379 |
| PubMedSearch : Amara_2012_Biochim.Biophys.Acta_1821_1379 |
| PubMedID: 22835523 |
| Gene_locus related to this paper: human-PNLIPRP2 |
| Title : High-performance liquid chromatography assay with fluorescence detection for the evaluation of inhibitors against human recombinant monoacylglycerol lipase - Holtfrerich_2010_Anal.Biochem_399_218 |
| Author(s) : Holtfrerich A , Makharadze T , Lehr M |
| Ref : Analytical Biochemistry , 399 :218 , 2010 |
| Abstract : Holtfrerich_2010_Anal.Biochem_399_218 |
| ESTHER : Holtfrerich_2010_Anal.Biochem_399_218 |
| PubMedSearch : Holtfrerich_2010_Anal.Biochem_399_218 |
| PubMedID: 20015447 |
| Title : Endocannabinoid biosynthesis proceeding through glycerophospho-N-acyl ethanolamine and a role for alpha\/beta-hydrolase 4 in this pathway - Simon_2006_J.Biol.Chem_281_26465 |
| Author(s) : Simon GM , Cravatt BF |
| Ref : Journal of Biological Chemistry , 281 :26465 , 2006 |
| Abstract : Simon_2006_J.Biol.Chem_281_26465 |
| ESTHER : Simon_2006_J.Biol.Chem_281_26465 |
| PubMedSearch : Simon_2006_J.Biol.Chem_281_26465 |
| PubMedID: 16818490 |
| Gene_locus related to this paper: human-ABHD4 |
| Title : Cannabinoid properties of methylfluorophosphonate analogs - Martin_2000_J.Pharmacol.Exp.Ther_294_1209 |
| Author(s) : Martin BR , Beletskaya I , Patrick G , Jefferson R , Winckler R , Deutsch DG , Di Marzo V , Dasse O , Mahadevan A , Razdan RK |
| Ref : Journal of Pharmacology & Experimental Therapeutics , 294 :1209 , 2000 |
| Abstract : Martin_2000_J.Pharmacol.Exp.Ther_294_1209 |
| ESTHER : Martin_2000_J.Pharmacol.Exp.Ther_294_1209 |
| PubMedSearch : Martin_2000_J.Pharmacol.Exp.Ther_294_1209 |
| PubMedID: 10945879 |