ML349
Quite specific for LYPLA2: APT2 (LYPLA2) Ki= 230 nM; APT1 (LYPLA1) Ki > 10microM
General
Type : Piperazine,Sulfur Compound
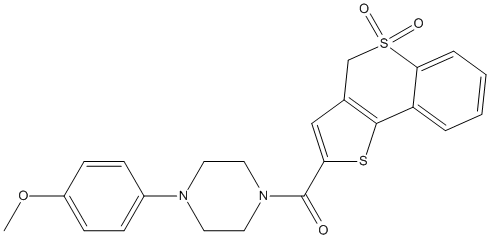
Chemical_Nomenclature : (5,5-dioxo-4H-thieno[3,2-c]thiochromen-2-yl)-[4-(4-methoxyphenyl)piperazin-1-yl]methanone
Canonical SMILES : COC1=CC=C(C=C1)N2CCN(CC2)C(=O)C3=CC4=C(S3)C5=CC=CC=C5S(=O)(=O)C4
InChI : InChI=1S\/C23H22N2O4S2\/c1-29-18-8-6-17(7-9-18)24-10-12-25(13-11-24)23(26)20-14-16-15-31(27,28)21-5-3-2-4-19(21)22(16)30-20\/h2-9,14H,10-13,15H2,1H3
InChIKey : YVIJPELUPZUEJX-UHFFFAOYSA-N
Other name(s) : 71T,ML 349,SMR000310192,SR-01000775306,1-[(5,5-dioxido-4H-thieno[3,2-c]thiochromen-2-yl)carbonyl]-4-(4-methoxyphenyl)piperazine
MW : 454.55
Formula : C23H22N2O4S2
CAS_number : 890819-86-0
PubChem : 16193817
UniChem : YVIJPELUPZUEJX-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : ML349 ligand of proteins in family: LYsophospholipase_carboxylesterase
Stucture : 5SYN Cocrystal structure of the human acyl protein thioesterase 2 with an isoform-selective inhibitor, ML349
Protein : human-LYPLA2
References (5)
| Title : Human leukocytes differentially express endocannabinoid-glycerol lipases and hydrolyze 2-arachidonoyl-glycerol and its metabolites from the 15-lipoxygenase and cyclooxygenase pathways - Turcotte_2019_J.Leukoc.Biol_106_1337 |
| Author(s) : Turcotte C , Dumais E , Archambault AS , Martin C , Blanchet MR , Bissonnette E , Boulet LP , Laviolette M , Di Marzo V , Flamand N |
| Ref : J Leukoc Biol , 106 :1337 , 2019 |
| Abstract : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| ESTHER : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| PubMedSearch : Turcotte_2019_J.Leukoc.Biol_106_1337 |
| PubMedID: 31556464 |
| Gene_locus related to this paper: human-ABHD6 , human-ABHD12 , human-ABHD16A , human-CES1 , human-LYPLA2 , human-PPT1 |
| Title : Molecular Mechanism for Isoform-Selective Inhibition of Acyl Protein Thioesterases 1 and 2 (APT1 and APT2) - Won_2016_ACS.Chem.Biol_11_3374 |
| Author(s) : Won SJ , Davda D , Labby KJ , Hwang SY , Pricer R , Majmudar JD , Armacost KA , Rodriguez LA , Rodriguez CL , Chong FS , Torossian KA , Palakurthi J , Hur ES , Meagher JL , Brooks CL, 3rd , Stuckey JA , Martin BR |
| Ref : ACS Chemical Biology , 11 :3374 , 2016 |
| Abstract : Won_2016_ACS.Chem.Biol_11_3374 |
| ESTHER : Won_2016_ACS.Chem.Biol_11_3374 |
| PubMedSearch : Won_2016_ACS.Chem.Biol_11_3374 |
| PubMedID: 27748579 |
| Gene_locus related to this paper: human-LYPLA2 |
| Title : Acyl protein thioesterase 1 and 2 (APT-1, APT-2) inhibitors palmostatin B, ML348 and ML349 have different effects on NRAS mutant melanoma cells - Vujic_2016_Oncotarget_7_7297 |
| Author(s) : Vujic I , Sanlorenzo M , Esteve-Puig R , Vujic M , Kwong A , Tsumura A , Murphy R , Moy A , Posch C , Monshi B , Rappersberger K , Ortiz-Urda S |
| Ref : Oncotarget , 7 :7297 , 2016 |
| Abstract : Vujic_2016_Oncotarget_7_7297 |
| ESTHER : Vujic_2016_Oncotarget_7_7297 |
| PubMedSearch : Vujic_2016_Oncotarget_7_7297 |
| PubMedID: 26771141 |
| Title : Acyl protein thioesterase inhibitors as probes of dynamic S-palmitoylation - Davda_2014_Medchemcomm_5_268 |
| Author(s) : Davda D , Martin BR |
| Ref : Medchemcomm , 5 :268 , 2014 |
| Abstract : Davda_2014_Medchemcomm_5_268 |
| ESTHER : Davda_2014_Medchemcomm_5_268 |
| PubMedSearch : Davda_2014_Medchemcomm_5_268 |
| PubMedID: 25558349 |
| Title : Characterization of a Selective, Reversible Inhibitor of Lysophospholipase 2 (LYPLA2) - Adibekian_2013_Probe.Report__4 |
| Author(s) : Adibekian A , Martin BR , Chang JW , Hsu KL , Tsuboi K , Bachovchin DA , Speers AE , Brown SJ , Spicer T , Fernandez-Vega V , Ferguson J , Cravatt BF , Hodder P , Rosen H |
| Ref : Probe Report , : , 2013 |
| Abstract : Adibekian_2013_Probe.Report__4 |
| ESTHER : Adibekian_2013_Probe.Report__4 |
| PubMedSearch : Adibekian_2013_Probe.Report__4 |
| PubMedID: 24624468 |