KT195
General
Type : Piperidine,Biphenyl,Triazol
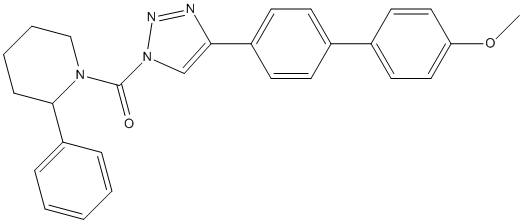
Chemical_Nomenclature : [4-[4-(4-methoxyphenyl)phenyl]triazol-1-yl]-(2-phenylpiperidin-1-yl)methanone
Canonical SMILES : COC1=CC=C(C=C1)C2=CC=C(C=C2)C3=CN(N=N3)C(=O)N4CCCCC4C5=CC=CC=C5
InChI : InChI=1S\/C27H26N4O2\/c1-33-24-16-14-21(15-17-24)20-10-12-22(13-11-20)25-19-31(29-28-25)27(32)30-18-6-5-9-26(30)23-7-3-2-4-8-23\/h2-4,7-8,10-17,19,26H,5-6,9,18H2,1H3
InChIKey : OEHLNQDRVMDXQD-UHFFFAOYSA-N
Other name(s) : ML295,(4-(4'-Methoxy-[1,1'-biphenyl]-4-yl)-1H-1,2,3-triazol-1-yl)(2-phenylpiperidin-1-yl)methanone,1402612-58-1,MLS004256815,CHEMBL3263571,SCHEMBL13192833,KT-195
MW : 438.52
Formula : C27H26N4O2
CAS_number :
PubChem : 53364533
UniChem : OEHLNQDRVMDXQD-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (4)
| Title : Regulation of calcium release from the endoplasmic reticulum by the serine hydrolase ABHD2 - Yun_2017_Biochem.Biophys.Res.Commun_490_1226 |
| Author(s) : Yun B , Lee H , Powell R , Reisdorph N , Ewing H , Gelb MH , Hsu KL , Cravatt BF , Leslie CC |
| Ref : Biochemical & Biophysical Research Communications , 490 :1226 , 2017 |
| Abstract : Yun_2017_Biochem.Biophys.Res.Commun_490_1226 |
| ESTHER : Yun_2017_Biochem.Biophys.Res.Commun_490_1226 |
| PubMedSearch : Yun_2017_Biochem.Biophys.Res.Commun_490_1226 |
| PubMedID: 28684316 |
| Gene_locus related to this paper: human-ABHD2 |
| Title : Comparative molecular field analysis and molecular dynamics studies of alpha\/beta hydrolase domain containing 6 (ABHD6) inhibitors - Kaczor_2015_J.Mol.Model_21_2789 |
| Author(s) : Kaczor AA , Targowska-Duda KM , Patel JZ , Laitinen T , Parkkari T , Adams Y , Nevalainen TJ , Poso A |
| Ref : J Mol Model , 21 :2789 , 2015 |
| Abstract : Kaczor_2015_J.Mol.Model_21_2789 |
| ESTHER : Kaczor_2015_J.Mol.Model_21_2789 |
| PubMedSearch : Kaczor_2015_J.Mol.Model_21_2789 |
| PubMedID: 26350245 |
| Title : Serine hydrolase inhibitors block necrotic cell death by preventing calcium overload of the mitochondria and permeability transition pore formation - Yun_2014_J.Biol.Chem_289_1491 |
| Author(s) : Yun B , Lee H , Ghosh M , Cravatt BF , Hsu KL , Bonventre JV , Ewing H , Gelb MH , Leslie CC |
| Ref : Journal of Biological Chemistry , 289 :1491 , 2014 |
| Abstract : Yun_2014_J.Biol.Chem_289_1491 |
| ESTHER : Yun_2014_J.Biol.Chem_289_1491 |
| PubMedSearch : Yun_2014_J.Biol.Chem_289_1491 |
| PubMedID: 24297180 |
| Title : DAGLbeta inhibition perturbs a lipid network involved in macrophage inflammatory responses - Hsu_2012_Nat.Chem.Biol_8_999 |
| Author(s) : Hsu KL , Tsuboi K , Adibekian A , Pugh H , Masuda K , Cravatt BF |
| Ref : Nat Chemical Biology , 8 :999 , 2012 |
| Abstract : Hsu_2012_Nat.Chem.Biol_8_999 |
| ESTHER : Hsu_2012_Nat.Chem.Biol_8_999 |
| PubMedSearch : Hsu_2012_Nat.Chem.Biol_8_999 |
| PubMedID: 23103940 |
| Gene_locus related to this paper: human-DAGLA , human-DAGLB |