H1R-7D9Q
General
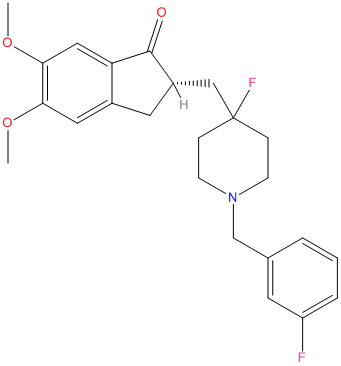
Type : Inden,Piperidine
Chemical_Nomenclature : (2S)-2-[[4-fluoranyl-1-[(3-fluorophenyl)methyl]piperidin-4-yl]methyl]-5,6-dimethoxy-2,3-dihydroinden-1-one
Canonical SMILES : COc1cc2c(cc1OC)C(=O)[C@@H](C2)CC3(CCN(CC3)Cc4cccc(c4)F)F
InChI : InChI=1S\/C24H27F2NO3\/c1-29-21-12-17-11-18(23(28)20(17)13-22(21)30-2)14-24(26)6-8-27(9-7-24)15-16-4-3-5-19(25)10-16\/h3-5,10,12-13,18H,6-9,11,14-15H2,1-2H3\/t18-\/m0\/s1
InChIKey : BVXQHSRGCUOBMQ-SFHVURJKSA-N
Other name(s) : H1R,Compound 7,(2~{S})-2-[[4-fluoranyl-1-[(3-fluorophenyl)methyl]piperidin-4-yl]methyl]-5,6-dimethoxy-2,3-dihydroinden-1-one
MW : 415.47
Formula : C24H27F2NO3
CAS_number :
PubChem :
UniChem : BVXQHSRGCUOBMQ-SFHVURJKSA-N
IUPHAR :
Wikipedia :
Target
Families : H1R-7D9Q ligand of proteins in family: ACHE
Stucture : 7D9Q Crystal Structure of Recombinant Human Acetylcholinesterase in Complex with Compound 7 H1R-7D9Q
Protein : human-ACHE
References (1)
| Title : Kinetics-Driven Drug Design Strategy for Next-Generation Acetylcholinesterase Inhibitors to Clinical Candidate - Zhou_2021_J.Med.Chem_64_1844 |
| Author(s) : Zhou Y , Fu Y , Yin W , Li J , Wang W , Bai F , Xu S , Gong Q , Peng T , Hong Y , Zhang D , Liu Q , Xu Y , Xu HE , Zhang H , Jiang H , Liu H |
| Ref : Journal of Medicinal Chemistry , 64 :1844 , 2021 |
| Abstract : Zhou_2021_J.Med.Chem_64_1844 |
| ESTHER : Zhou_2021_J.Med.Chem_64_1844 |
| PubMedSearch : Zhou_2021_J.Med.Chem_64_1844 |
| PubMedID: 33570950 |
| Gene_locus related to this paper: human-ACHE |