GSK237826A
General
Type : Piperidine
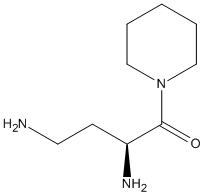
Chemical_Nomenclature : (2S)-2,4-diamino-1-piperidin-1-ylbutan-1-one
Canonical SMILES : C1CCN(CC1)C(=O)C(CCN)N
InChI : InChI=1S\/C9H19N3O\/c10-5-4-8(11)9(13)12-6-2-1-3-7-12\/h8H,1-7,10-11H2\/t8-\/m0\/s1
InChIKey : RKBKYSFKXFKBBM-QMMMGPOBSA-N
Other name(s) : (3S)-4-oxo-4-piperidin-1-ylbutane-1,3-diamine,OPY,Dab-Pip,DPP II Inhibitor,CHEMBL93505,Dipeptidylpeptidase II Inhibitor,L-2,4-Diaminobutyrylpiperidinamide,L-2,4-diaminobutrylpiperidinamide
MW : 185.27
Formula : C9H19N3O
CAS_number :
PubChem : 9942336
UniChem : RKBKYSFKXFKBBM-QMMMGPOBSA-N
IUPHAR :
Wikipedia :
Target
Families : GSK237826A ligand of proteins in family: Prolylcarboxypeptidase
Stucture : 3N0T Human dipeptidyl peptidase DPP7 complexed with inhibitor GSK237826A
Protein : human-DPP7
References (5)
| Title : Crystal structure and substrate recognition mechanism of the prolyl endoprotease PEP from Aspergillus niger - Miyazono_2022_Biochem.Biophys.Res.Commun_591_76 |
| Author(s) : Miyazono KI , Kubota K , Takahashi K , Tanokura M |
| Ref : Biochemical & Biophysical Research Communications , 591 :76 , 2022 |
| Abstract : Miyazono_2022_Biochem.Biophys.Res.Commun_591_76 |
| ESTHER : Miyazono_2022_Biochem.Biophys.Res.Commun_591_76 |
| PubMedSearch : Miyazono_2022_Biochem.Biophys.Res.Commun_591_76 |
| PubMedID: 34999257 |
| Gene_locus related to this paper: aspnc-a2qr21 |
| Title : Structures of human DPP7 reveal the molecular basis of specific inhibition and the architectural diversity of proline-specific peptidases - Bezerra_2012_PLoS.One_7_e43019 |
| Author(s) : Bezerra GA , Dobrovetsky E , Dong A , Seitova A , Crombett L , Shewchuk LM , Hassell AM , Sweitzer SM , Sweitzer TD , McDevitt PJ , Johanson KO , Kennedy-Wilson KM , Cossar D , Bochkarev A , Gruber K , Dhe-Paganon S |
| Ref : PLoS ONE , 7 :e43019 , 2012 |
| Abstract : Bezerra_2012_PLoS.One_7_e43019 |
| ESTHER : Bezerra_2012_PLoS.One_7_e43019 |
| PubMedSearch : Bezerra_2012_PLoS.One_7_e43019 |
| PubMedID: 22952628 |
| Gene_locus related to this paper: human-DPP7 |
| Title : In vivo effects of a potent, selective DPPII inhibitor: UAMC00039 is a possible tool for the elucidation of the physiological function of DPPII - |
| Author(s) : Maes MB , Lambeir AM , Van der Veken P , De Winter B , Augustyns K , Scharpe S , De Meester I |
| Ref : Advances in Experimental Medicine & Biology , 575 :73 , 2006 |
| PubMedID: 16700510 |
| Title : Peptide substrates of dipeptidyl peptidases - |
| Author(s) : Brandt I , Lambeir AM , Maes MB , Scharpe S , De Meester I |
| Ref : Advances in Experimental Medicine & Biology , 575 :3 , 2006 |
| PubMedID: 16700503 |
| Title : Design, synthesis, and SAR of potent and selective dipeptide-derived inhibitors for dipeptidyl peptidases - Senten_2003_J.Med.Chem_46_5005 |
| Author(s) : Senten K , Van der Veken P , De Meester I , Lambeir AM , Scharpe S , Haemers A , Augustyns K |
| Ref : Journal of Medicinal Chemistry , 46 :5005 , 2003 |
| Abstract : Senten_2003_J.Med.Chem_46_5005 |
| ESTHER : Senten_2003_J.Med.Chem_46_5005 |
| PubMedSearch : Senten_2003_J.Med.Chem_46_5005 |
| PubMedID: 14584950 |