Desoxypeganine
Alkaloid from plants of the Zygophyllaceae family Inhibits both cholinesterases and monoamine oxydases.Inhibits butyrylcholinesterase more strongly than acetylcholinesterase. Desoxypeganine hydrochloride crosses easily the blood-brain barrier.
General
Type : Not A\/B H target,Alkaloid,Natural,Quinazoline
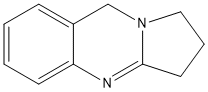
Chemical_Nomenclature : 1,2,3,9-tetrahydropyrrolo[2,1-b]quinazoline
Canonical SMILES : C1CC2=NC3=CC=CC=C3CN2C1
InChI : InChI=1S\/C11H12N2\/c1-2-5-10-9(4-1)8-13-7-3-6-11(13)12-10\/h1-2,4-5H,3,6-8H2
InChIKey : WUFQLZTXIWKION-UHFFFAOYSA-N
Other name(s) : Deoxypeganin,Deoxypeganine,3-Deoxyvasicine,ZINC00265524,C10656,Pegene hydrochloride,Pyrrolo(2,1-b)quinazoline, 1,2,3,9-tetrahydro-
MW : 245
Formula : C11H12N2HCl 2H2O
CAS_number : 61939-05-7 || 459-59-0
PubChem : 442894
UniChem : WUFQLZTXIWKION-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (2)
| Title : Differential inhibition of [3H]-oxotremorine-M and [3H]-quinuclinidyl benzilate binding to muscarinic receptors in rat brain membranes with acetylcholinesterase inhibitors - Lockhart_2001_Naunyn.Schmiedebergs.Arch.Pharmacol_363_429 |
| Author(s) : Lockhart B , Closier M , Howard K , Steward C , Lestage P |
| Ref : Naunyn Schmiedebergs Arch Pharmacol , 363 :429 , 2001 |
| Abstract : Lockhart_2001_Naunyn.Schmiedebergs.Arch.Pharmacol_363_429 |
| ESTHER : Lockhart_2001_Naunyn.Schmiedebergs.Arch.Pharmacol_363_429 |
| PubMedSearch : Lockhart_2001_Naunyn.Schmiedebergs.Arch.Pharmacol_363_429 |
| PubMedID: 11330337 |
| Title : [Pharmacological characteristics of desoxypeganine hydrochloride] - Tuliaganov_1986_Farmakol.Toksikol_49_37 |
| Author(s) : Tuliaganov N , Sadritdinov FS , Suleimanova GA |
| Ref : Farmakologiia i Toksikologiia , 49 :37 , 1986 |
| Abstract : Tuliaganov_1986_Farmakol.Toksikol_49_37 |
| ESTHER : Tuliaganov_1986_Farmakol.Toksikol_49_37 |
| PubMedSearch : Tuliaganov_1986_Farmakol.Toksikol_49_37 |
| PubMedID: 3720932 |