Debromoflustramine-B
Debromofluustramine B (176), a prenylated pyrrolidinoindolinefound in the bryozoan Flustra foliacea. (-)-2 enantiomer (CID 10403152) has the lowest IC50 1.37 muM
General
Type : Natural,Pyrrolidine,Indole,Derivative of physostigmine-eserine
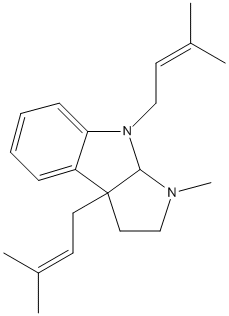
Chemical_Nomenclature : 3-methyl-4,8b-bis(3-methylbut-2-enyl)-2,3a-dihydro-1H-pyrrolo[2,3-b]indole
Canonical SMILES : CC(=CCC12CCN(C1N(C3=CC=CC=C23)CC=C(C)C)C)C
InChI : InChI=1S\/C21H30N2\/c1-16(2)10-12-21-13-15-22(5)20(21)23(14-11-17(3)4)19-9-7-6-8-18(19)21\/h6-11,20H,12-15H2,1-5H3
InChIKey : NUWYFFDZPIGJJO-UHFFFAOYSA-N || NUWYFFDZPIGJJO-RTWAWAEBSA-N
Other name(s) : CHEMBL452582,NSC676585,AC1LAVLJ,BDBM50245239,CHEMBL383307,BDBM50241051
MW : 310.48
Formula : C21H30N2
CAS_number :
UniChem : NUWYFFDZPIGJJO-UHFFFAOYSA-N || NUWYFFDZPIGJJO-RTWAWAEBSA-N
IUPHAR :
Wikipedia :
Target
References (3)
| Title : Natural cholinesterase inhibitors from marine organisms - Moodie_2019_Nat.Prod.Rep_36_1053 |
| Author(s) : Moodie LWK , Sepcic K , Turk T , Frange ZR , Svenson J |
| Ref : Nat Prod Rep , 36 :1053 , 2019 |
| Abstract : Moodie_2019_Nat.Prod.Rep_36_1053 |
| ESTHER : Moodie_2019_Nat.Prod.Rep_36_1053 |
| PubMedSearch : Moodie_2019_Nat.Prod.Rep_36_1053 |
| PubMedID: 30924818 |
| Title : In silico ligand-receptor docking of potentially selective butyrylcholinesterase inhibitors structurally related to the marine natural product debromoflustramine B - Mitchell_2010_Med.Chem_6_141 |
| Author(s) : Mitchell MO , Figliozzi RW , Guzel M |
| Ref : Med Chem , 6 :141 , 2010 |
| Abstract : Mitchell_2010_Med.Chem_6_141 |
| ESTHER : Mitchell_2010_Med.Chem_6_141 |
| PubMedSearch : Mitchell_2010_Med.Chem_6_141 |
| PubMedID: 20632975 |
| Title : Synthesis and biological evaluation of (-)- and (+)-debromoflustramine B and its analogues as selective butyrylcholinesterase inhibitors - Rivera-Becerril_2008_J.Med.Chem_51_5271 |
| Author(s) : Rivera-Becerril E , Joseph-Nathan P , Perez-Alvarez VM , Morales-Rios MS |
| Ref : Journal of Medicinal Chemistry , 51 :5271 , 2008 |
| Abstract : Rivera-Becerril_2008_J.Med.Chem_51_5271 |
| ESTHER : Rivera-Becerril_2008_J.Med.Chem_51_5271 |
| PubMedSearch : Rivera-Becerril_2008_J.Med.Chem_51_5271 |
| PubMedID: 18686941 |