Cpd37-Fluoroindol-carbamate
IC50 muM FAAH (not A/B hydrolase) 0.038; MAGL 0.038; BChE 2.3; AChE inactive
General
Type : Multitarget,Carbamate,Indole,FAAH inhibitor,Alkyl linked bis-ligand
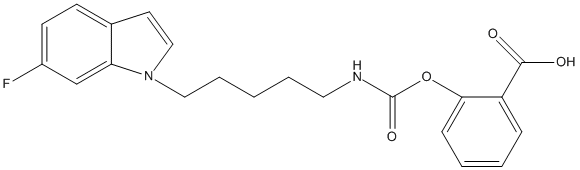
Chemical_Nomenclature : 2-({[5-(6-Fluoroindol-1-yl)pentyl]carbamoyl}oxy)benzoic Acid
Canonical SMILES : C(CCCCN(C(=O)OC1=CC=CC=C1C(=O)O)[H])[N]3C2=CC(=CC=C2C=C3)F
InChI : InChI=1S\/C21H21FN2O4\/c22-16-9-8-15-10-13-24(18(15)14-16)12-5-1-4-11-23-21(27)28-19-7-3-2-6-17(19)20(25)26\/h2-3,6-10,13-14H,1,4-5,11-12H2,(H,23,27)(H,25,26)
InChIKey : KCGOKRVIXFMPPS-UHFFFAOYSA-N
Other name(s) :
MW : 384.40
Formula : C21H21FN2O4
CAS_number :
PubChem :
UniChem : KCGOKRVIXFMPPS-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : Cpd37-Fluoroindol-carbamate ligand of proteins in family: Monoglyceridelipase_lysophospholip
Stucture :
Protein : human-MGLL
References (1)
| Title : Aryl N-[w-(6-Fluoroindol-1-yl)alkyl]carbamates as Inhibitors of Fatty Acid Amide Hydrolase, Monoacylglycerol Lipase, and Butyrylcholinesterase: Structure-Activity Relationships and Hydrolytic Stability - Rudolph_2021_ACS.Omega_6_13466 |
| Author(s) : Rudolph S , Dahlhaus H , Hanekamp W , Albers C , Barth M , Michels G , Friedrich D , Lehr M |
| Ref : ACS Omega , 6 :13466 , 2021 |
| Abstract : Rudolph_2021_ACS.Omega_6_13466 |
| ESTHER : Rudolph_2021_ACS.Omega_6_13466 |
| PubMedSearch : Rudolph_2021_ACS.Omega_6_13466 |
| PubMedID: 34056494 |