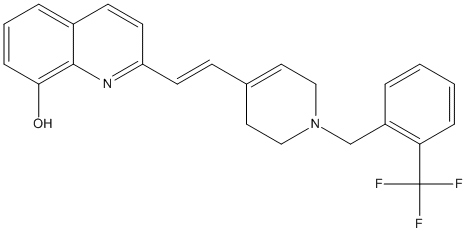
Compound19n-quinolinol
IC50 AChE 161 nM
General
Type : Trifluo,anti-Abeta-aggregation,Metal ion chelator,Quinoline,Multitarget,Derivative of Donepezil
Chemical_Nomenclature : (E)-2-(2-(1-(2-(trifluoromethylbenzyl))-1,2,3,6-tetrahydropyridin-4-yl)vinyl)quinolin-8ol hydrochloride
Canonical SMILES : C1=CC(=C2C(=C1)C=CC(=N2)C=CC3=CCN(CC3)CC4=C(C=CC=C4)C(F)(F)F)O
InChI : InChI=1S\/C24H21F3N2O\/c25-24(26,27)21-6-2-1-4-19(21)16-29-14-12-17(13-15-29)8-10-20-11-9-18-5-3-7-22(30)23(18)28-20\/h1-12,30H,13-16H2\/b10-8+
InChIKey : HCUMFCRMHUJZMD-CSKARUKUSA-N
Other name(s) :
MW : 410.43
Formula : C24H21F3N2O
CAS_number :
PubChem :
UniChem : HCUMFCRMHUJZMD-CSKARUKUSA-N
IUPHAR :
Wikipedia :
Target
References (1)
| Title : Discovery of novel hybrids containing clioquinol-1-benzyl-1,2,3,6-tetrahydropyridine as multi-target-directed ligands (MTDLs) against Alzheimer's disease - Li_2022_Eur.J.Med.Chem_244_114841 |
| Author(s) : Li X , Li T , Zhang P , Lu L , Sun Y , Zhang B , Allen S , White L , Phillips J , Zhu Z , Yao H , Xu J |
| Ref : Eur Journal of Medicinal Chemistry , 244 :114841 , 2022 |
| Abstract : Li_2022_Eur.J.Med.Chem_244_114841 |
| ESTHER : Li_2022_Eur.J.Med.Chem_244_114841 |
| PubMedSearch : Li_2022_Eur.J.Med.Chem_244_114841 |
| PubMedID: 36257284 |