CHEMBL2152538
Formed after carbamoylation of BChE's active site by carbamate inhibitors such as: Tetracyclic-carbamate-cp8. Strongly reduced toxicity on hippocampal neuronal cells (ht-22). IC50 AChE 110.3 microM BChE 44.3 microM
General
Type : Not A\/B H target,Isoquinoline,Quinazoline
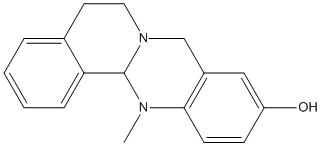
Chemical_Nomenclature : 13-methyl-5,6,8,13a-tetrahydroisoquinolino[1,2-b]quinazolin-10-ol
Canonical SMILES : CN1C2C3=CC=CC=C3CCN2CC4=C1C=CC(=C4)O
InChI : InChI=1S\/C17H18N2O\/c1-18-16-7-6-14(20)10-13(16)11-19-9-8-12-4-2-3-5-15(12)17(18)19\/h2-7,10,17,20H,8-9,11H2,1H3
InChIKey : YMWULAJABIPEAC-UHFFFAOYSA-N
Other name(s) : SCHEMBL22239399,TQR1174,BDBM50393001,13-Methyl-5,8,13,13a-tetrahydro-6H-isoquino[1,2-b]quinazoline-10-ol
MW : 266.34
Formula : C17H18N2O
CAS_number :
PubChem : 71456586
UniChem : YMWULAJABIPEAC-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families :
Stucture :
Protein :
References (2)
| Title : Selective Pseudo-irreversible Butyrylcholinesterase Inhibitors Transferring Antioxidant Moieties to the Enzyme Show Pronounced Neuroprotective Efficacy In Vitro and In Vivo in an Alzheimer's Disease Mouse Model - Scheiner_2021_J.Med.Chem_64_9302 |
| Author(s) : Scheiner M , Hoffmann M , He F , Poeta E , Chatonnet A , Monti B , Maurice T , Decker M |
| Ref : Journal of Medicinal Chemistry , 64(13): :9302 , 2021 |
| Abstract : Scheiner_2021_J.Med.Chem_64_9302 |
| ESTHER : Scheiner_2021_J.Med.Chem_64_9302 |
| PubMedSearch : Scheiner_2021_J.Med.Chem_64_9302 |
| PubMedID: 34152756 |
| Title : Neuroprotective Tri- and Tetracyclic BChE Inhibitors Releasing Reversible Inhibitors upon Carbamate Transfer - Darras_2012_ACS.Med.Chem.Lett_3_914 |
| Author(s) : Darras FH , Kling B , Heilmann J , Decker M |
| Ref : ACS Med Chem Lett , 3 :914 , 2012 |
| Abstract : Darras_2012_ACS.Med.Chem.Lett_3_914 |
| ESTHER : Darras_2012_ACS.Med.Chem.Lett_3_914 |
| PubMedSearch : Darras_2012_ACS.Med.Chem.Lett_3_914 |
| PubMedID: 24900407 |