CHEMBL2147710
General
Type : Trifluoro,Pyrrolidine,Thiazolidine,Sulfur Compound,Pyrazole
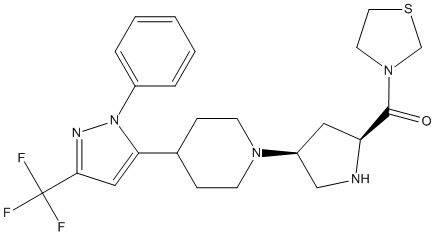
Chemical_Nomenclature : [(2S,4S)-4-[4-[2-phenyl-5-(trifluoromethyl)pyrazol-3-yl]piperidin-1-yl]pyrrolidin-2-yl]-(1,3-thiazolidin-3-yl)methanone
Canonical SMILES : C1CN(CCC1C2=CC(=NN2C3=CC=CC=C3)C(F)(F)F)C4CC(NC4)C(=O)N5CCSC5
InChI : InChI=1S\/C23H28F3N5OS\/c24-23(25,26)21-13-20(31(28-21)17-4-2-1-3-5-17)16-6-8-29(9-7-16)18-12-19(27-14-18)22(32)30-10-11-33-15-30\/h1-5,13,16,18-19,27H,6-12,14-15H2\/t18-,19-\/m0\/s1
InChIKey : CYNIHEXFNGMKLP-OALUTQOASA-N
Other name(s) : CHEMBL2147710,BDBM50391574,[(2s,4s)-4-{4-[1-Phenyl-3-(Trifluoromethyl)-1h-Pyrazol-5-Yl]piperidin-1-Yl}pyrrolidin-2-Yl](1,3-Thiazolidin-3-Yl)methanone,W94
MW : 479.56
Formula : C23H28F3N5OS
CAS_number :
PubChem : 66545741
UniChem : CYNIHEXFNGMKLP-OALUTQOASA-N
IUPHAR :
Wikipedia :
Target
Families : CHEMBL2147710 ligand of proteins in family: DPP4N_Peptidase_S9
Stucture : 3VJL Crystal structure of human dipeptidyl peptidase IV (DPP-4) in complex with a prolylthiazolidine inhibitor 2
Protein : human-DPP4
References (4)
| Title : Discovery and preclinical profile of teneligliptin (3-[(2S,4S)-4-[4-(3-methyl-1-phenyl-1H-pyrazol-5-yl)piperazin-1-yl]pyrrolidin-2-y lcarbonyl]thiazolidine): a highly potent, selective, long-lasting and orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes - Yoshida_2012_Bioorg.Med.Chem_20_5705 |
| Author(s) : Yoshida T , Akahoshi F , Sakashita H , Kitajima H , Nakamura M , Sonda S , Takeuchi M , Tanaka Y , Ueda N , Sekiguchi S , Ishige T , Shima K , Nabeno M , Abe Y , Anabuki J , Soejima A , Yoshida K , Takashina Y , Ishii S , Kiuchi S , Fukuda S , Tsutsumiuchi R , Kosaka K , Murozono T , Nakamaru Y , Utsumi H , Masutomi N , Kishida H , Miyaguchi I , Hayashi Y |
| Ref : Bioorganic & Medicinal Chemistry , 20 :5705 , 2012 |
| Abstract : Yoshida_2012_Bioorg.Med.Chem_20_5705 |
| ESTHER : Yoshida_2012_Bioorg.Med.Chem_20_5705 |
| PubMedSearch : Yoshida_2012_Bioorg.Med.Chem_20_5705 |
| PubMedID: 22959556 |
| Gene_locus related to this paper: human-DPP4 |
| Title : Fused bicyclic heteroarylpiperazine-substituted L-prolylthiazolidines as highly potent DPP-4 inhibitors lacking the electrophilic nitrile group - Yoshida_2012_Bioorg.Med.Chem_20_5033 |
| Author(s) : Yoshida T , Akahoshi F , Sakashita H , Sonda S , Takeuchi M , Tanaka Y , Nabeno M , Kishida H , Miyaguchi I , Hayashi Y |
| Ref : Bioorganic & Medicinal Chemistry , 20 :5033 , 2012 |
| Abstract : Yoshida_2012_Bioorg.Med.Chem_20_5033 |
| ESTHER : Yoshida_2012_Bioorg.Med.Chem_20_5033 |
| PubMedSearch : Yoshida_2012_Bioorg.Med.Chem_20_5033 |
| PubMedID: 22824762 |
| Gene_locus related to this paper: human-DPP4 |
| Title : [(S)-gamma-(4-Aryl-1-piperazinyl)-l-prolyl]thiazolidines as a novel series of highly potent and long-lasting DPP-IV inhibitors - Yoshida_2007_Bioorg.Med.Chem.Lett_17_2618 |
| Author(s) : Yoshida T , Sakashita H , Akahoshi F , Hayashi Y |
| Ref : Bioorganic & Medicinal Chemistry Lett , 17 :2618 , 2007 |
| Abstract : Yoshida_2007_Bioorg.Med.Chem.Lett_17_2618 |
| ESTHER : Yoshida_2007_Bioorg.Med.Chem.Lett_17_2618 |
| PubMedSearch : Yoshida_2007_Bioorg.Med.Chem.Lett_17_2618 |
| PubMedID: 17317162 |
| Gene_locus related to this paper: human-DPP4 |
| Title : [(S)-gamma-(Arylamino)prolyl]thiazolidine compounds as a novel series of potent and stable DPP-IV inhibitors - Sakashita_2006_Bioorg.Med.Chem_14_3662 |
| Author(s) : Sakashita H , Akahoshi F , Kitajima H , Tsutsumiuchi R , Hayashi Y |
| Ref : Bioorganic & Medicinal Chemistry , 14 :3662 , 2006 |
| Abstract : Sakashita_2006_Bioorg.Med.Chem_14_3662 |
| ESTHER : Sakashita_2006_Bioorg.Med.Chem_14_3662 |
| PubMedSearch : Sakashita_2006_Bioorg.Med.Chem_14_3662 |
| PubMedID: 16460948 |
| Gene_locus related to this paper: human-DPP4 |