Bis3-tacrine
rat brain AChE IC50 (nM) 254+/-55; rat serum BuChE IC50(nM) 152+/-17
General
Type : Derivative of Tacrine,Alkyl linked bis-ligand,Acridine
Chemical_Nomenclature : N,N'-bis(1,2,3,4-tetrahydroacridin-9-yl)propane-1,3-diamine
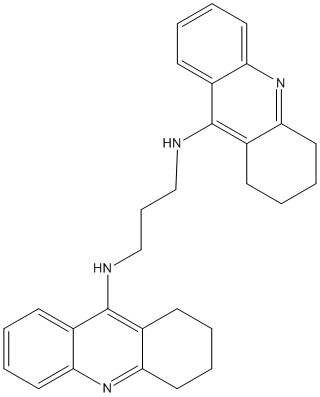
Canonical SMILES : C1CCC2=NC3=CC=CC=C3C(=C2C1)NCCCNC4=C5CCCCC5=NC6=CC=CC=C64
InChI : InChI=1S\/C29H32N4\/c1-5-14-24-20(10-1)28(21-11-2-6-15-25(21)32-24)30-18-9-19-31-29-22-12-3-7-16-26(22)33-27-17-8-4-13-23(27)29\/h1,3,5,7,10,12,14,16H,2,4,6,8-9,11,13,15,17-19H2,(H,30,32)(H,31,33)
InChIKey : BHOCVLCAWPPEOV-UHFFFAOYSA-N
Other name(s) : B3C,bis(3)-cognitin,bis(3)-tacrine,C577051,CHEMBL378006,Bis-THA inhibitor 2,A3A.2HCl,SCHEMBL13480729,BDBM10479
MW :
Formula : C29H32N4
CAS_number :
PubChem : 12069888
UniChem : BHOCVLCAWPPEOV-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (6)
| Title : Tacrine induces endoplasmic reticulum-stressed apoptosis via disrupting the proper assembly of oligomeric acetylcholinesterase in cultured neuronal cells - Liu_2021_Mol.Pharmacol__ |
| Author(s) : Liu EYL , Mak S , Kong X , Xia Y , Kwan KKL , Xu ML , Tsim KWK |
| Ref : Molecular Pharmacology , : , 2021 |
| Abstract : Liu_2021_Mol.Pharmacol__ |
| ESTHER : Liu_2021_Mol.Pharmacol__ |
| PubMedSearch : Liu_2021_Mol.Pharmacol__ |
| PubMedID: 34531295 |
| Title : Photopharmacology on Acetylcholinesterase: Novel Photoswitchable Inhibitors with Improved Pharmacological Profiles - Scheiner_2021_ChemPhotoChem_5_149 |
| Author(s) : Scheiner M , Sink A , Spatz P , Endres E , Decker M |
| Ref : ChemPhotoChem , 5 :149 , 2021 |
| Abstract : Scheiner_2021_ChemPhotoChem_5_149 |
| ESTHER : Scheiner_2021_ChemPhotoChem_5_149 |
| PubMedSearch : Scheiner_2021_ChemPhotoChem_5_149 |
| PubMedID: |
| Title : Promising tacrine\/huperzine A-based dimeric acetylcholinesterase inhibitors for neurodegenerative disorders: From relieving symptoms to modifying diseases through multitarget - Mak_2021_J.Neurochem__ |
| Author(s) : Mak S , Li W , Fu H , Luo J , Cui W , Hu S , Pang Y , Carlier PR , Tsim KWK , Pi R , Han Y |
| Ref : Journal of Neurochemistry , : , 2021 |
| Abstract : Mak_2021_J.Neurochem__ |
| ESTHER : Mak_2021_J.Neurochem__ |
| PubMedSearch : Mak_2021_J.Neurochem__ |
| PubMedID: 33930191 |
| Title : Activation of transcription factor MEF2D by bis(3)-cognitin protects dopaminergic neurons and ameliorates Parkinsonian motor defects - Yao_2012_J.Biol.Chem_287_34246 |
| Author(s) : Yao L , Li W , She H , Dou J , Jia L , He Y , Yang Q , Zhu J , Capiro NL , Walker DI , Pennell KD , Pang Y , Liu Y , Han Y , Mao Z |
| Ref : Journal of Biological Chemistry , 287 :34246 , 2012 |
| Abstract : Yao_2012_J.Biol.Chem_287_34246 |
| ESTHER : Yao_2012_J.Biol.Chem_287_34246 |
| PubMedSearch : Yao_2012_J.Biol.Chem_287_34246 |
| PubMedID: 22891246 |
| Title : Search of antitubercular activities in tetrahydroacridines: synthesis and biological evaluation - Tripathi_2006_Bioorg.Med.Chem.Lett_16_5144 |
| Author(s) : Tripathi RP , Verma SS , Pandey J , Agarwal KC , Chaturvedi V , Manju YK , Srivastva AK , Gaikwad A , Sinha S |
| Ref : Bioorganic & Medicinal Chemistry Lett , 16 :5144 , 2006 |
| Abstract : Tripathi_2006_Bioorg.Med.Chem.Lett_16_5144 |
| ESTHER : Tripathi_2006_Bioorg.Med.Chem.Lett_16_5144 |
| PubMedSearch : Tripathi_2006_Bioorg.Med.Chem.Lett_16_5144 |
| PubMedID: 16870429 |
| Title : Complexes of alkylene-linked tacrine dimers with Torpedo californica acetylcholinesterase: Binding of Bis5-tacrine produces a dramatic rearrangement in the active-site gorge - Rydberg_2006_J.Med.Chem_49_5491 |
| Author(s) : Rydberg EH , Brumshtein B , Greenblatt HM , Wong DM , Shaya D , Williams LD , Carlier PR , Pang YP , Silman I , Sussman JL |
| Ref : Journal of Medicinal Chemistry , 49 :5491 , 2006 |
| Abstract : Rydberg_2006_J.Med.Chem_49_5491 |
| ESTHER : Rydberg_2006_J.Med.Chem_49_5491 |
| PubMedSearch : Rydberg_2006_J.Med.Chem_49_5491 |
| PubMedID: 16942022 |
| Gene_locus related to this paper: torca-ACHE |