ARUK3005518
General
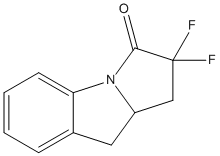
Type : Fragment inhibitor of Notum,Indole
Chemical_Nomenclature : (3~{a}~{S})-2,2-bis(fluoranyl)-3~{a},4-dihydro-3~{H}-pyrrolo[1,2-a]indol-1-one
Canonical SMILES : c1ccc2c(c1)C[C@@H]3N2C(=O)C(C3)(F)F || [C]1=[C][C]=C2C(=[C]1)N3[C](C2)CC(C3=O)(F)F
InChI : InChI=1S\/C11H9F2NO\/c12-11(13)6-8-5-7-3-1-2-4-9(7)14(8)10(11)15\/h1-4,8H,5-6H2\/t8-\/m0\/s1
InChIKey : FBNLACZWDFCRLD-QMMMGPOBSA-N
Other name(s) : RGO,Compound 5m
MW : 209.19
Formula : C11H9F2NO
CAS_number :
PubChem :
UniChem : FBNLACZWDFCRLD-QMMMGPOBSA-N
IUPHAR :
Wikipedia :
Target
Families : ARUK3005518 ligand of proteins in family: Pectinacetylesterase-Notum
Stucture : 8BT0 Structure of the human Wnt deacylase Notum in complex with ARUK3005518
Protein : human-NOTUM
References (1)
| Title : Designed switch from covalent to non-covalent inhibitors of carboxylesterase Notum activity - Atkinson_2023_Eur.J.Med.Chem_251_115132 |
| Author(s) : Atkinson BN , Willis NJ , Zhao Y , Patel C , Frew S , Costelloe K , Magno L , Svensson F , Jones EY , Fish PV |
| Ref : Eur Journal of Medicinal Chemistry , 251 :115132 , 2023 |
| Abstract : Atkinson_2023_Eur.J.Med.Chem_251_115132 |
| ESTHER : Atkinson_2023_Eur.J.Med.Chem_251_115132 |
| PubMedSearch : Atkinson_2023_Eur.J.Med.Chem_251_115132 |
| PubMedID: 36934521 |
| Gene_locus related to this paper: human-NOTUM |