Hypaphorine
Tryptophan betaine, fungal auxin antagonist. Betaines are synthesized or taken up from the environment by cells for protection against osmotic stress, drought, high salinity, or high temperature. Zwitterions quaternary ammonium or phosphonium cation that bears no hydrogen atom and with a negatively charged functional group not adjacent to the cationic site. Not proper alkaloid but often classified as such.Attenuates Lipopolysaccharide-Induced Endothelial Inflammation, induces sleep
General
Type : Not A\/B H target,Natural,Derivative of Tryptophan,Indole,Propionate
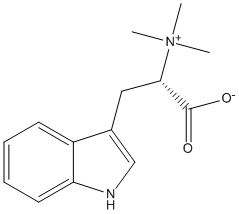
Chemical_Nomenclature : (2S)-3-(1H-indol-3-yl)-2-(trimethylazaniumyl)propanoate
Canonical SMILES : C[N+](C)(C)C(CC1=CNC2=CC=CC=C21)C(=O)[O-]
InChI : InChI=1S\/C14H18N2O2\/c1-16(2,3)13(14(17)18)8-10-9-15-12-7-5-4-6-11(10)12\/h4-7,9,13,15H,8H2,1-3H3\/t13-\/m0\/s1
InChIKey : AOHCBEAZXHZMOR-ZDUSSCGKSA-N
Other name(s) : Lenticin,L-Hypaphorine,Tryptophan betaine,Glyyunnanenine,(+)-Hypaphorine,CHEBI:5832,C14H18N2O2,GJ3358U63L,Hypaforin,Lenticine,L-tryptophan betaine,SCHEMBL1460893,CHEMBL2235164,DL-Hypaphorine,3-(1H-indol-3-yl)-2-(trimethylazaniumyl)propanoate,Hypaphorine chloride,SCHEMBL22099726
MW : 246.30
Formula : C14H18N2O2
CAS_number : 487-58-1
PubChem : 442106, 3861164, 24189566
UniChem : AOHCBEAZXHZMOR-ZDUSSCGKSA-N
IUPHAR :
Wikipedia :
Target
References (1)
| Title : l-Hypaphorine and d-hypaphorine: Specific antiacetylcholinesterase activity in rat brain tissue - Yonekawa_2021_Bioorg.Med.Chem.Lett_47_128206 |
| Author(s) : Yonekawa MKA , Penteado BB , Dal'Ongaro Rodrigues A , Lourenco EMG , Barbosa EG , das Neves SC , de Oliveira RJ , Marques MR , Silva DB , de Lima DP , Beatriz A , Oses JP , Dos SJJA , Santos E |
| Ref : Bioorganic & Medicinal Chemistry Lett , 47 :128206 , 2021 |
| Abstract : Yonekawa_2021_Bioorg.Med.Chem.Lett_47_128206 |
| ESTHER : Yonekawa_2021_Bioorg.Med.Chem.Lett_47_128206 |
| PubMedSearch : Yonekawa_2021_Bioorg.Med.Chem.Lett_47_128206 |
| PubMedID: 34146704 |