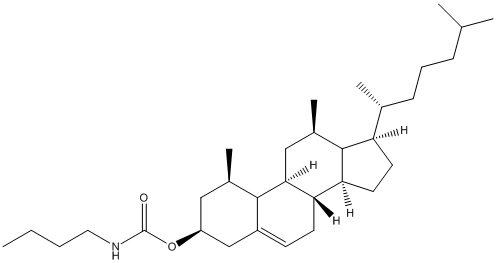
3-Cholesteryl-N-butylcarbamate
General
Type : Carbamate,Steroid,Phenanthrene,Lipase inhibitor
Chemical_Nomenclature : [(3S,8S,9S,10R,13R,14S,17R)-10,13-dimethyl-17-[(2R)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl] N-butylcarbamate
Canonical SMILES : CCCCNC(=O)OC1CCC2(C3CCC4(C(C3CC=C2C1)CCC4C(C)CCCC(C)C)C)C
InChI : InChI=1S\/C32H55NO2\/c1-7-8-20-33-30(34)35-25-16-18-31(5)24(21-25)12-13-26-28-15-14-27(23(4)11-9-10-22(2)3)32(28,6)19-17-29(26)31\/h12,22-23,25-29H,7-11,13-21H2,1-6H3,(H,33,34)\/t23-,25+,26+,27-,28+,29+,31+,32-\/m1\/s1
InChIKey : YRLNNPIPHGFZON-PTHRTHQKSA-N
Other name(s) : 3-Cholesteryl N-butylcarbamate,3-Cnbc,DTXSID50967290,Cholest-5-en-3-ol (3beta)-, butylcarbamate,Cholest-5-en-3-yl hydrogen butylcarbonimidate
MW : 485.8
Formula : C32H55NO2
CAS_number : 52829-26-2
PubChem : 6452731
UniChem : YRLNNPIPHGFZON-PTHRTHQKSA-N
IUPHAR :
Wikipedia :
Target
Families : 3-Cholesteryl-N-butylcarbamate ligand of proteins in family: Cholesterol_esterase
Stucture :
Protein : pig-i3ltk9
References (1)
| Title : p-Nitrophenyl and cholesteryl-N-alkyl carbamates as inhibitors of cholesterol esterase - Hosie_1987_J.Biol.Chem_262_260 |
| Author(s) : Hosie L , Sutton LD , Quinn DM |
| Ref : Journal of Biological Chemistry , 262 :260 , 1987 |
| Abstract : Hosie_1987_J.Biol.Chem_262_260 |
| ESTHER : Hosie_1987_J.Biol.Chem_262_260 |
| PubMedSearch : Hosie_1987_J.Biol.Chem_262_260 |
| PubMedID: 3793726 |
| Gene_locus related to this paper: pig-i3ltk9 |