t-MTUCB-NH
Comparable cytotoxicity to sorafenib, but without inhibitory activity against the primary targets of sorafenib, such as C-Raf kinase and vascular endothelial growth factor receptor 2 (VEGFR2) kinase
General
Type : Urea derivative,Multitarget
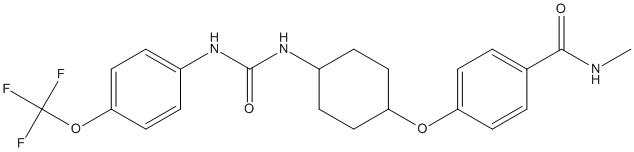
Chemical_Nomenclature : N-Methyl-4-{[trans-4-({[4-(trifluoromethoxy)phenyl]carbamoyl}amino)cyclohexyl]oxy}benzamide
Canonical SMILES : CNC(=O)C1=CC=C(C=C1)OC2CCC(CC2)NC(=O)NC3=CC=C(C=C3)OC(F)(F)F
InChI : InChI=1S\/C22H24F3N3O4\/c1-26-20(29)14-2-8-17(9-3-14)31-18-10-4-15(5-11-18)27-21(30)28-16-6-12-19(13-7-16)32-22(23,24)25\/h2-3,6-9,12-13,15,18H,4-5,10-11H2,1H3,(H,26,29)(H2,27,28,30)
InChIKey : JNPWHXLRBKEIPP-UHFFFAOYSA-N
Other name(s) : CHEMBL2397138,CHEMBL3353249,CHEMBL3895613,SCHEMBL15271834
MW : 451.4
Formula : C22H24F3N3O4
CAS_number :
PubChem : 60196738
UniChem : JNPWHXLRBKEIPP-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : t-MTUCB-NH ligand of proteins in family: Epoxide_hydrolase
Stucture :
Protein : human-EPHX2 || mouse-hyes
References (1)
| Title : Novel sorafenib-based structural analogues: in-vitro anticancer evaluation of t-MTUCB and t-AUCMB - Wecksler_2014_Anticancer.Drugs_25_433 |
| Author(s) : Wecksler AT , Hwang SH , Wettersten HI , Gilda JE , Patton A , Leon LJ , Carraway KL, 3rd , Gomes AV , Baar K , Weiss RH , Hammock BD |
| Ref : Anticancer Drugs , 25 :433 , 2014 |
| Abstract : Wecksler_2014_Anticancer.Drugs_25_433 |
| ESTHER : Wecksler_2014_Anticancer.Drugs_25_433 |
| PubMedSearch : Wecksler_2014_Anticancer.Drugs_25_433 |
| PubMedID: 24525589 |