Piperidine-constrained-phenethylamine-42
General
Type : Trifluoro,Sulfur Compound,Pyrimidine,Piperidine
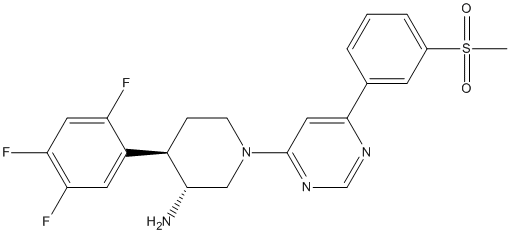
Chemical_Nomenclature : (3R,4R)-1-[6-(3-methylsulfonylphenyl)pyrimidin-4-yl]-4-(2,4,5-trifluorophenyl)piperidin-3-amine
Canonical SMILES : CS(=O)(=O)C1=CC=CC(=C1)C2=CC(=NC=N2)N3CCC(C(C3)N)C4=CC(=C(C=C4F)F)F
InChI : InChI=1S\/C22H21F3N4O2S\/c1-32(30,31)14-4-2-3-13(7-14)21-10-22(28-12-27-21)29-6-5-15(20(26)11-29)16-8-18(24)19(25)9-17(16)23\/h2-4,7-10,12,15,20H,5-6,11,26H2,1H3\/t15-,20+\/m1\/s1
InChIKey : GOBIXGZJSMAOFV-QRWLVFNGSA-N
Other name(s) : MA9,piperidine-constrained phenethylamine,CHEMBL1234200,BDBM15518,DB08164,Piperidine-Constrained Phenethylamine 42,(3R,4R)-1-(6-(3-(methylsulfonyl)phenyl)pyrimidin-4-yl)-4-(2,4,5-trifluorophenyl)-piperidin-3-amine
MW : 462.49
Formula : C22H21F3N4O2S
CAS_number :
PubChem : 16058650
UniChem : GOBIXGZJSMAOFV-QRWLVFNGSA-N
IUPHAR :
Wikipedia :
Target
Families : Piperidine-constrained-phenethylamine-42 ligand of proteins in family: DPP4N_Peptidase_S9
Stucture : 2OQV Human Dipeptidyl Peptidase IV (DPP4) with piperidine-constrained phenethylamine
Protein : human-DPP4
References (1)
| Title : Discovery and structure-activity relationships of piperidinone- and piperidine-constrained phenethylamines as novel, potent, and selective dipeptidyl peptidase IV inhibitors - Pei_2007_J.Med.Chem_50_1983 |
| Author(s) : Pei Z , Li X , von Geldern TW , Longenecker K , Pireh D , Stewart KD , Backes BJ , Lai C , Lubben TH , Ballaron SJ , Beno DW , Kempf-Grote AJ , Sham HL , Trevillyan JM |
| Ref : Journal of Medicinal Chemistry , 50 :1983 , 2007 |
| Abstract : Pei_2007_J.Med.Chem_50_1983 |
| ESTHER : Pei_2007_J.Med.Chem_50_1983 |
| PubMedSearch : Pei_2007_J.Med.Chem_50_1983 |
| PubMedID: 17367123 |
| Gene_locus related to this paper: human-DPP4 |