Phenothiazine-Tacrine-1dC
1dC is a potent and selective acetylcholinesterase inhibitor with IC50 = 8 nM (1aA whithout chlore and shorter linker potent BChE inhibitor with IC50 = 15 nM )
General
Type : Phenothiazine,Multitarget,Derivative of Tacrine,Antioxidant,Acridine,Alkyl linked bis-ligand,anti-Abeta-aggregation,Sulfur Compound
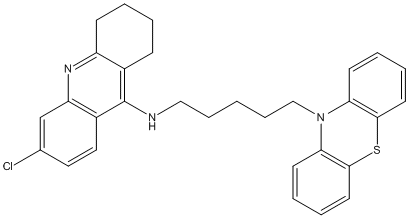
Chemical_Nomenclature : 6-chloro-N-[5-(10H-phenothiazin-10-yl)pentyl]-1,2,3,4-tetrahydroacridin-9-amine
Canonical SMILES : C1=C(C=CC2=C1N=C6C(=C2NCCCCCN3C5=C(SC4=C3C=CC=C4)C=CC=C5)CCCC6)Cl
InChI : InChI=1S\/C30H30ClN3S\/c31-21-16-17-23-25(20-21)33-24-11-3-2-10-22(24)30(23)32-18-8-1-9-19-34-26-12-4-6-14-28(26)35-29-15-7-5-13-27(29)34\/h4-7,12-17,20H,1-3,8-11,18-19H2,(H,32,33)
InChIKey : ICNPKYKIVQPEDC-UHFFFAOYSA-N
Other name(s) :
Target
Families : Phenothiazine-Tacrine-1dC ligand of proteins in family
ACHE
Protein :
human-ACHE
References (1)
| Title : Phenothiazine-Tacrine Heterodimers: Pursuing Multitarget Directed Approach in Alzheimer's Disease - Gorecki_2021_ACS.Chem.Neurosci__ |
| Author(s) : Gorecki L , Uliassi E , Bartolini M , Janockova J , Hrabinova M , Hepnarova V , Prchal L , Muckova L , Pejchal J , Karasova JZ , Mezeiova E , Benkova M , Kobrlova T , Soukup O , Petralla S , Monti B , Korabecny J , Bolognesi ML |
| Ref : ACS Chem Neurosci , : , 2021 |
| Abstract : Gorecki_2021_ACS.Chem.Neurosci__ |
| ESTHER : Gorecki_2021_ACS.Chem.Neurosci__ |
| PubMedSearch : Gorecki_2021_ACS.Chem.Neurosci__ |
| PubMedID: 33852284 |