IMP-DDAO
Sarin analogue. Fluorogenic organophosphate diesters that produce fluorescence emission upon hydrolysis. The first ester is ethyl (E), isopropyl (I), cyclohexyl (C) or pinacoyl (P) analogous to the structure of VX, Sarin, Cyclosarin, and Soman respectively. The fluorescent ester is 3-cyano-4-methyl-7-hydroxy coumarin (MeCyC) or 1,3-dichloro-7-hydroxy-9,9-dimethyl-9H-acridin-2-one (DDAO)
General
Type : Organophosphate,Flu-OP,Nerve Agent G-series,Surrogate OP,Derivative of Tacrine,Derivative of Sarin
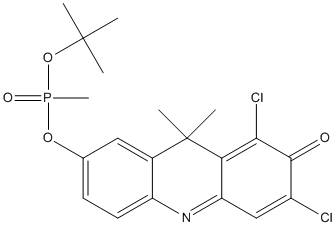
Chemical_Nomenclature : methylphosphonic acid isopropyl ester 6,8-dichloro-9,9-dimethyl-7-oxo-7,9-dihydro-acridin-2-yl
Canonical SMILES : CC2(C1=C(C=CC(=C1)O[P](=O)(C)OC(C)(C)C)N=C3C2=C(C(=O)C(=C3)Cl)Cl)C
InChI : InChI=1S\/C20H22Cl2NO4P\/c1-19(2,3)27-28(6,25)26-11-7-8-14-12(9-11)20(4,5)16-15(23-14)10-13(21)18(24)17(16)22\/h7-10H,1-6H3
InChIKey : OLUSHKKVVWQKFX-UHFFFAOYSA-N
Other name(s) :
MW : 442.27
Formula : C20H22Cl2NO4P
CAS_number :
PubChem :
UniChem : OLUSHKKVVWQKFX-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (2)
| Title : Asymmetric fluorogenic organophosphates for the development of active organophosphate hydrolases with reversed stereoselectivity - Amitai_2007_Toxicology_233_187 |
| Author(s) : Amitai G , Adani R , Yacov G , Yishay S , Teitlboim S , Tveria L , Limanovich O , Kushnir M , Meshulam H |
| Ref : Toxicology , 233 :187 , 2007 |
| Abstract : Amitai_2007_Toxicology_233_187 |
| ESTHER : Amitai_2007_Toxicology_233_187 |
| PubMedSearch : Amitai_2007_Toxicology_233_187 |
| PubMedID: 17129656 |
| Title : Enhanced stereoselective hydrolysis of toxic organophosphates by directly evolved variants of mammalian serum paraoxonase - Amitai_2006_FEBS.J_273_1906 |
| Author(s) : Amitai G , Gaidukov L , Adani R , Yishay S , Yacov G , Kushnir M , Teitlboim S , Lindenbaum M , Bel P , Khersonsky O , Tawfik DS , Meshulam H |
| Ref : Febs J , 273 :1906 , 2006 |
| Abstract : Amitai_2006_FEBS.J_273_1906 |
| ESTHER : Amitai_2006_FEBS.J_273_1906 |
| PubMedSearch : Amitai_2006_FEBS.J_273_1906 |
| PubMedID: 16640555 |