ABC47
potent dual inhibitor of ABHD3 and ABHD4 (IC50 values of 0.13 and 0.03 M, respectively)
General
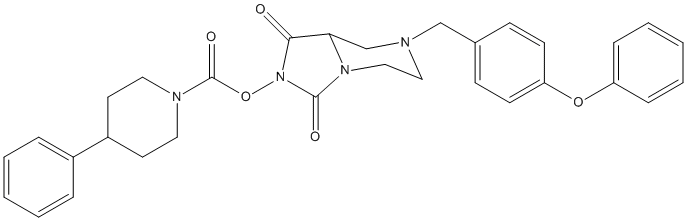
Type : Carbamate,Piperidine,N-hydroxyhydantoin,Phenoxyphenyl
Chemical_Nomenclature : [1,3-dioxo-7-[(4-phenoxyphenyl)methyl]-5,6,8,8a-tetrahydroimidazo[1,5-a]pyrazin-2-yl] 4-phenylpiperidine-1-carboxylate
Canonical SMILES : C1CN(CCC1C2=CC=CC=C2)C(=O)ON3C(=O)C4CN(CCN4C3=O)CC5=CC=C(C=C5)OC6=CC=CC=C6
InChI : InChI=1S\/C31H32N4O5\/c36-29-28-22-32(21-23-11-13-27(14-12-23)39-26-9-5-2-6-10-26)19-20-34(28)30(37)35(29)40-31(38)33-17-15-25(16-18-33)24-7-3-1-4-8-24\/h1-14,25,28H,15-22H2
InChIKey : RZRRRWRXKABFDO-UHFFFAOYSA-N
Other name(s) : SCHEMBL17275314,ABC-47
MW : 540.6
Formula : C31H32N4O5
CAS_number :
PubChem : 118525585
UniChem : RZRRRWRXKABFDO-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : ABC47 ligand of proteins in family: abh_upf0017 || CGI-58_ABHD5_ABHD4
Stucture :
Protein : human-ABHD3 || human-ABHD4
References (1)
| Title : Selective N-Hydroxyhydantoin Carbamate Inhibitors of Mammalian Serine Hydrolases - Cognetta_2015_Chem.Biol_22_928 |
| Author(s) : Cognetta AB, 3rd , Niphakis MJ , Lee HC , Martini ML , Hulce JJ , Cravatt BF |
| Ref : Chemical Biology , 22 :928 , 2015 |
| Abstract : Cognetta_2015_Chem.Biol_22_928 |
| ESTHER : Cognetta_2015_Chem.Biol_22_928 |
| PubMedSearch : Cognetta_2015_Chem.Biol_22_928 |
| PubMedID: 26120000 |