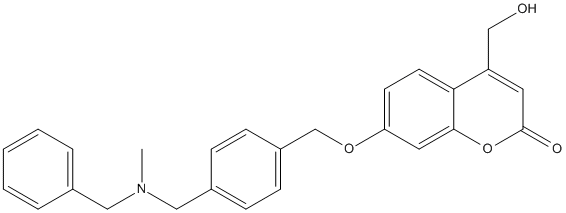
7QAK-Cpd1
MAO-B Ki muM 4.5 +/-0.2; MAO-B IC50 muM 0.010 +/- 0.002; human AChE IC50 muM 0.12 +/- 0.01; mouse AChE IC50 muM 0.40 +/- 0.03
General
Type : Monoamine-oxidase-inhibitor,Multitarget,Coumarin
Chemical_Nomenclature : 7-[(4-{[benzyl(methyl)amino]methyl}benzyl)oxy]-4-(hydroxymethyl)-2H-chromen-2-one || 4-Hydroxymethyl-7-(4-(methylbenzylaminomethyl)benzyloxy)-2H-1-benzopyran-2-one
Canonical SMILES : CN(CC1=CC=CC=C1)CC2=CC=C(C=C2)COC3=CC4=C(C=C3)C(=CC(=O)O4)CO
InChI : InChI=1S\/C26H25NO4\/c1-27(15-19-5-3-2-4-6-19)16-20-7-9-21(10-8-20)18-30-23-11-12-24-22(17-28)13-26(29)31-25(24)14-23\/h2-14,28H,15-18H2,1H3
InChIKey : RTJRBYKZQSYWCM-UHFFFAOYSA-N
Other name(s) : CHEMBL3586771
MW : 415.5
Formula : C26H25NO4
CAS_number :
PubChem : 122180463
UniChem : RTJRBYKZQSYWCM-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
References (1)
| Title : Dual Reversible Coumarin Inhibitors Mutually Bound to Monoamine Oxidase B and Acetylcholinesterase Crystal Structures - Ekstrom_2022_ACS.Med.Chem.Lett_13_499 |
| Author(s) : Ekstrom F , Gottinger A , Forsgren N , Catto M , Iacovino LG , Pisani P , Binda C |
| Ref : ACS Med Chem Lett , 13 :499 , 2022 |
| Abstract : Ekstrom_2022_ACS.Med.Chem.Lett_13_499 |
| ESTHER : Ekstrom_2022_ACS.Med.Chem.Lett_13_499 |
| PubMedSearch : Ekstrom_2022_ACS.Med.Chem.Lett_13_499 |
| PubMedID: 35300078 |
| Gene_locus related to this paper: mouse-ACHE |